A Bomb Calorimeter Is Used To Measure
Juapaving
Apr 05, 2025 · 6 min read
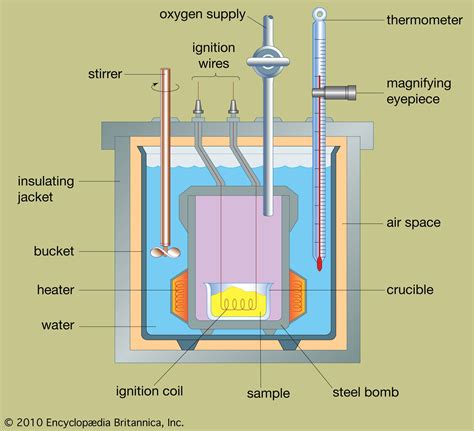
Table of Contents
A Bomb Calorimeter is Used to Measure: The Ultimate Guide to Calorimetry
A bomb calorimeter, also known as a constant volume calorimeter, is a crucial instrument used in numerous scientific fields to precisely measure the heat of combustion of a substance. Understanding its function and applications is essential for anyone working with thermodynamics, chemistry, and related disciplines. This comprehensive guide will delve into the intricacies of bomb calorimetry, explaining its principle, operation, applications, and limitations.
Understanding the Principle of Bomb Calorimetry
At its core, a bomb calorimeter operates on the principle of conservation of energy. The heat released during the combustion of a sample is completely absorbed by the calorimeter and its contents, leading to a measurable temperature increase. This temperature change, combined with the calorimeter's heat capacity, allows for the calculation of the heat of combustion, often expressed in kJ/mol or kcal/mol.
The process involves placing a precisely weighed sample within a robust, sealed container – the "bomb" – filled with high-pressure oxygen. The bomb is then submerged in a water bath, ensuring that the heat generated during combustion is transferred efficiently to the surrounding water. The temperature of the water is meticulously monitored before, during, and after the combustion process.
Key Components of a Bomb Calorimeter:
- Bomb: A strong, airtight metal container designed to withstand the high pressure and temperature generated during combustion.
- Ignition System: An electrical system used to initiate the combustion process.
- Water Bath: A well-insulated container filled with a known volume of water, acting as a heat sink.
- Thermometer/Temperature Sensor: A highly sensitive device used to precisely measure the temperature change of the water bath.
- Stirrer: A mechanism designed to ensure uniform temperature distribution within the water bath.
The Combustion Process and Data Acquisition
The combustion process itself is rapid and exothermic, meaning it releases heat. The heat released by the sample is absorbed by the calorimeter, resulting in a measurable temperature increase in the water bath. This increase is directly proportional to the heat of combustion of the substance.
Steps Involved in a Bomb Calorimetry Experiment:
- Sample Preparation: A precisely weighed sample is carefully placed within the bomb. The sample should be thoroughly dried to minimize errors.
- Bomb Assembly: The bomb is sealed and filled with high-pressure oxygen to ensure complete combustion.
- Calorimeter Setup: The bomb is carefully submerged in the water bath, ensuring complete immersion.
- Ignition: The combustion process is initiated using the electrical ignition system.
- Temperature Monitoring: The temperature of the water bath is monitored continuously using a high-precision thermometer or temperature sensor. Data is typically recorded at regular intervals before, during, and after the combustion.
- Data Analysis: The temperature change is used, along with the heat capacity of the calorimeter, to calculate the heat of combustion using appropriate thermodynamic equations.
Calculating the Heat of Combustion
The heat of combustion (ΔH<sub>c</sub>) is calculated using the following formula:
ΔH<sub>c</sub> = -C<sub>cal</sub> * ΔT / n
Where:
- ΔH<sub>c</sub> is the heat of combustion (usually expressed in kJ/mol or kcal/mol).
- C<sub>cal</sub> is the heat capacity of the calorimeter (determined through calibration using a standard substance with a known heat of combustion, such as benzoic acid).
- ΔT is the change in temperature of the water bath.
- n is the number of moles of the sample.
The negative sign indicates that the combustion process is exothermic (releases heat). The heat capacity of the calorimeter, C<sub>cal</sub>, is a crucial parameter that needs to be determined accurately through calibration experiments.
Applications of Bomb Calorimetry
Bomb calorimetry finds extensive applications in various fields, including:
1. Food Science and Nutrition:
Bomb calorimetry is widely used to determine the caloric content of foods and nutritional supplements. By accurately measuring the heat of combustion of a food sample, nutritionists can determine its energy value.
2. Environmental Science:
In environmental studies, bomb calorimetry is employed to assess the energy content of fuels and waste materials. This information is crucial in designing efficient waste management strategies and evaluating the energy potential of different fuels.
3. Chemical Engineering:
In chemical engineering, bomb calorimetry helps determine the heat of reaction for various chemical processes. This information is vital for designing efficient and safe chemical reactors.
4. Forensic Science:
Bomb calorimetry can be used in forensic investigations to analyze the energy content of explosive materials. This information can aid in identifying the type of explosive used and reconstructing the events surrounding an explosion.
5. Materials Science:
Researchers in materials science utilize bomb calorimetry to characterize the energetic properties of new materials. This data is critical in evaluating the suitability of new materials for various applications.
Limitations of Bomb Calorimetry
Despite its wide range of applications, bomb calorimetry has certain limitations:
- Incomplete Combustion: If the sample doesn't completely combust, the measured heat of combustion will be underestimated. Ensuring sufficient oxygen pressure and a finely divided sample are crucial to minimizing this issue.
- Formation of Unstable Products: The formation of unstable or reactive products during combustion can lead to errors in the measurements.
- Calibration Challenges: Accurate calibration of the calorimeter is crucial for obtaining reliable results. Any errors in the calibration process will propagate into the heat of combustion calculation.
- Specialized Equipment: Bomb calorimetry requires specialized and expensive equipment, which might not be accessible to all researchers.
- Sample Size Limitations: The technique might not be suitable for very small or very large samples. The sample size must be carefully chosen to ensure accurate and reliable results.
Advanced Techniques and Future Trends
Recent advancements in bomb calorimetry include the use of more sophisticated temperature sensors, automated data acquisition systems, and improved software for data analysis. These advancements have significantly enhanced the accuracy and precision of bomb calorimetry measurements.
Future trends in bomb calorimetry may involve the integration of advanced analytical techniques, such as mass spectrometry and gas chromatography, to provide a more comprehensive understanding of the combustion process. The development of miniaturized and portable bomb calorimeters could also expand the applicability of this technique to a wider range of settings.
Conclusion
Bomb calorimetry remains a powerful and versatile technique for measuring the heat of combustion of various substances. Its wide-ranging applications across diverse scientific fields highlight its importance in understanding the energetic properties of materials and processes. While limitations exist, advancements in instrumentation and data analysis continue to enhance the accuracy and reliability of this essential technique. By understanding the principles, procedures, and limitations of bomb calorimetry, researchers can harness its power to advance knowledge and solve critical problems in various scientific disciplines.
Latest Posts
Latest Posts
-
What Is The Scientific Name For A Cat
Apr 06, 2025
-
How Many Valence Electrons In Iodine
Apr 06, 2025
-
How Many Feet In 3 Yard
Apr 06, 2025
-
Find An Eigenvector Corresponding To The Eigenvalue
Apr 06, 2025
-
Which Of The Following Is Correct
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about A Bomb Calorimeter Is Used To Measure . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
