Does Higher Electronegativity Mean Stronger Acid
Juapaving
Apr 03, 2025 · 5 min read
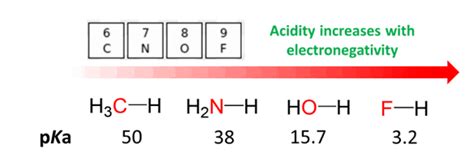
Table of Contents
Does Higher Electronegativity Mean Stronger Acid? A Comprehensive Exploration
The relationship between electronegativity and acid strength is a fundamental concept in chemistry, often simplified to the statement: "higher electronegativity means stronger acid." While this generalization holds true in many cases, it's crucial to understand the nuances and exceptions to this rule for a comprehensive understanding. This article delves deep into the complexities of this relationship, exploring various factors influencing acid strength and providing a clear picture of when electronegativity plays a dominant role and when it doesn't.
Understanding Electronegativity and its Influence on Acidity
Electronegativity, a crucial concept in chemistry, measures an atom's ability to attract electrons within a chemical bond. Atoms with high electronegativity strongly pull electrons towards themselves, creating a polar bond if bonded to a less electronegative atom. This electron-pulling power significantly impacts the acidity of a molecule.
How Electronegativity Affects Bond Polarity and Acid Strength
In the context of acids (molecules that donate protons, H+), the electronegativity of the atom bonded to the acidic hydrogen (H) plays a vital role. A highly electronegative atom draws electron density away from the O-H bond, weakening the bond and making it easier to release a proton (H+). This leads to a stronger acid.
Example: Consider the series of hydrohalic acids (HF, HCl, HBr, HI). Fluorine (F) is the most electronegative element. However, HF is a weaker acid than HCl, HBr, and HI. This apparent contradiction highlights that electronegativity is not the sole determinant of acid strength. Bond strength and other factors play significant roles.
The Role of Bond Strength in Acid Strength
The strength of the bond between the hydrogen atom and the atom it's attached to (often oxygen or a halogen) is inversely proportional to the acid strength. A weaker bond means the proton is more easily released, resulting in a stronger acid. While a highly electronegative atom can weaken the bond, the overall bond strength is affected by factors beyond just electronegativity.
Example: The large size of iodine in HI leads to a longer and weaker H-I bond compared to the shorter and stronger H-F bond in HF. This weaker bond in HI makes it a much stronger acid than HF despite fluorine's higher electronegativity.
Factors Beyond Electronegativity Affecting Acid Strength
While electronegativity contributes significantly, other crucial factors influence acid strength, often overshadowing the effects of electronegativity alone. These factors include:
1. Bond Length and Bond Strength:
As discussed earlier, longer and weaker bonds are associated with stronger acids. This is because less energy is required to break the bond and release the proton. This effect often outweighs the impact of electronegativity, as seen in the hydrohalic acids.
2. Resonance Stabilization:
Resonance stabilization is a powerful factor influencing acid strength. If the conjugate base (the species remaining after the acid donates a proton) is resonance-stabilized, it's more stable, and the acid is stronger. Resonance delocalizes the negative charge, making it less concentrated and more stable.
Example: Carboxylic acids are stronger acids than alcohols due to the resonance stabilization of their conjugate base (carboxylate ion). The negative charge is delocalized over two oxygen atoms, making the conjugate base more stable.
3. Inductive Effect:
The inductive effect describes the electron-withdrawing or electron-donating ability of substituents attached to the molecule. Electron-withdrawing groups stabilize the negative charge on the conjugate base, increasing the acid's strength. Electron-donating groups have the opposite effect, weakening the acid.
Example: The presence of electronegative atoms (like chlorine) near the carboxyl group in chloroacetic acid increases its acidity compared to acetic acid. The chlorine atom inductively withdraws electrons, stabilizing the conjugate base.
4. Hybridization:
The hybridization of the atom bonded to the acidic hydrogen also affects acid strength. Atoms with higher s-character (like sp hybridized carbons) hold electrons more tightly, resulting in weaker acids. Conversely, atoms with lower s-character (like sp3 hybridized carbons) are less electronegative, leading to stronger acids.
5. Solvent Effects:
The solvent in which the acid is dissolved plays a critical role in determining its apparent strength. The solvent can interact with the acid and its conjugate base, influencing the equilibrium and thus the acidity. Protic solvents (solvents with O-H or N-H bonds) can stabilize both the acid and its conjugate base through hydrogen bonding, affecting the overall acidity.
Exceptions to the Rule: When Higher Electronegativity Doesn't Mean Stronger Acid
The statement "higher electronegativity means stronger acid" is a simplification. Numerous examples contradict this generalization.
Hydrohalic Acids (HF, HCl, HBr, HI):
As highlighted earlier, despite fluorine's highest electronegativity, HF is the weakest hydrohalic acid. The exceptionally strong H-F bond outweighs the effect of fluorine's high electronegativity.
Comparing Acids with Different Anions:
Comparing acids with different anions (the negatively charged part of the acid) requires a careful consideration of all the factors mentioned above. Electronegativity might play a role, but other factors like resonance stabilization and inductive effects frequently dominate.
Conclusion: A Holistic Perspective on Acid Strength
While electronegativity is a significant factor influencing acid strength, it's not the sole determinant. A comprehensive understanding of acid strength requires considering several interacting factors, including bond strength, resonance stabilization, inductive effects, hybridization, and solvent effects. The relationship between electronegativity and acid strength is complex and multifaceted. In many cases, other factors outweigh the effect of electronegativity, leading to exceptions to the general rule. A holistic approach is necessary to accurately predict and explain the relative acidity of different molecules. Therefore, while electronegativity provides valuable insights, it should be treated as one piece of the puzzle rather than the complete solution when determining acid strength. Understanding these nuances is vital for a thorough grasp of acid-base chemistry.
Latest Posts
Latest Posts
-
Difference Between Spongy And Compact Bone
Apr 04, 2025
-
D How Is The Energy Produced By Respiration Stored
Apr 04, 2025
-
The Functional And Structural Unit Of The Kidneys Is The
Apr 04, 2025
-
To Pour Water On Calcium Oxide
Apr 04, 2025
-
Evaluate The Trigonometric Function At The Quadrantal Angle
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Does Higher Electronegativity Mean Stronger Acid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
