Difference Between Localized And Delocalized Electrons
Juapaving
Apr 03, 2025 · 6 min read
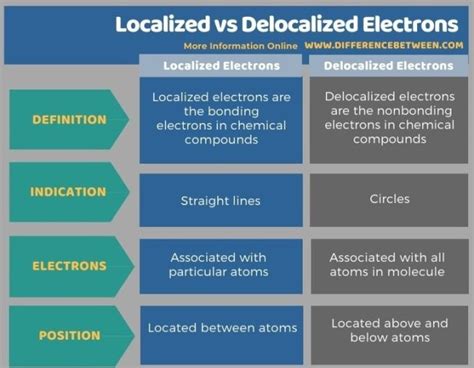
Table of Contents
Delocalized vs. Localized Electrons: A Comprehensive Guide
Understanding the difference between localized and delocalized electrons is crucial for grasping fundamental concepts in chemistry, particularly in organic chemistry and materials science. This distinction impacts a molecule's properties, reactivity, and overall behavior. This article delves deep into the concept, explaining the core differences, providing examples, and exploring the implications of electron delocalization.
What are Localized Electrons?
Localized electrons are electrons that are associated with a specific atom or a particular covalent bond within a molecule. They are confined to a relatively small region of space and are not free to move throughout the entire molecule. Think of them as being "stuck" in their position.
Characteristics of Localized Electrons:
- Restricted Movement: Localized electrons are primarily found within the atomic orbitals of individual atoms or within the bonding orbitals of a specific covalent bond. Their movement is largely restricted to this area.
- Stronger Bonds: The localized nature of electrons in covalent bonds leads to stronger, more defined bonds. The electron density is concentrated between the bonded atoms.
- Predictable Reactivity: Molecules with predominantly localized electrons exhibit more predictable reactivity patterns, as the electron density is concentrated in specific locations.
- Examples: Most simple organic molecules like methane (CH₄), ethane (C₂H₆), and many saturated hydrocarbons exhibit predominantly localized electrons. The electrons in the C-H and C-C sigma bonds are localized between the bonded atoms.
Visualizing Localized Electrons:
Imagine drawing a Lewis structure. The electrons represented by lone pairs or those participating in single, double, or triple bonds are considered localized. Each electron pair is directly associated with a specific atom or bond.
What are Delocalized Electrons?
Delocalized electrons are electrons that are not associated with a single atom or covalent bond but are instead spread out or shared among several atoms or molecules. They are free to move throughout a larger region of space, often across the entire molecule.
Characteristics of Delocalized Electrons:
- Free Movement: Delocalized electrons are not confined to a specific atom or bond; they are mobile and can occupy several atomic or molecular orbitals simultaneously.
- Weaker Individual Bonds: While the overall molecule is often more stable due to delocalization, the individual bonds may be slightly weaker than localized bonds because electron density is spread over a larger area.
- Increased Stability: Delocalization leads to increased stability of the molecule due to resonance. This enhanced stability is reflected in lower energy and reduced reactivity.
- Unusual Reactivity: Molecules with delocalized electrons may exhibit less predictable reactivity patterns compared to those with only localized electrons, as the electron density is spread across a larger area, creating different reactive sites.
- Examples: Benzene (C₆H₆) is a classic example. The six π electrons in the benzene ring are delocalized, creating a ring current. Other examples include conjugated systems (alternating single and multiple bonds), aromatic compounds, and certain metal complexes.
Visualizing Delocalized Electrons:
Delocalized electrons are best represented by resonance structures. In benzene, for example, the double and single bonds are not fixed but instead represent the average distribution of electron density across the ring. The actual structure is a hybrid of all resonance contributors, showing the delocalized π electron cloud above and below the ring.
Key Differences Between Localized and Delocalized Electrons:
| Feature | Localized Electrons | Delocalized Electrons |
|---|---|---|
| Location | Associated with a specific atom or bond | Spread over several atoms or molecules |
| Movement | Restricted to a small region | Free to move over a larger region |
| Bond Strength | Stronger individual bonds | Slightly weaker individual bonds, stronger overall |
| Stability | Less stable than molecules with delocalized electrons | More stable due to resonance |
| Reactivity | More predictable reactivity | Less predictable, potentially more reactive in specific areas |
| Representation | Single Lewis structure | Multiple resonance structures, showing electron cloud |
Implications of Delocalization:
Delocalization has profound implications on the properties of molecules:
- Increased Stability: Resonance stabilization, resulting from delocalization, significantly lowers the molecule's energy. This increased stability translates to lower reactivity.
- Enhanced Color: Delocalized electrons often interact with light in the visible spectrum, leading to colored compounds. Many dyes and pigments rely on extensive delocalization.
- Electrical Conductivity: In some materials, delocalized electrons contribute to electrical conductivity. Metals, for example, have a "sea" of delocalized electrons responsible for their conductive properties.
- Magnetic Properties: Delocalized electrons can lead to paramagnetism or diamagnetism, depending on the electron configuration and molecular structure.
- Spectroscopic Properties: Delocalized electrons influence the absorption and emission spectra of molecules, making them valuable tools for analysis.
Examples Illustrating the Difference:
1. Ethane (C₂H₆) vs. Benzene (C₆H₆):
Ethane has only localized electrons in its C-C and C-H sigma bonds. Benzene, on the other hand, possesses six delocalized π electrons forming a stable aromatic ring. This difference leads to a vast difference in reactivity. Ethane readily undergoes free-radical substitution reactions, while benzene undergoes electrophilic aromatic substitution, a far more specific and less reactive process.
2. Carbon Dioxide (CO₂) vs. Ozone (O₃):
In carbon dioxide, the electrons are localized in double bonds between carbon and oxygen atoms. Ozone, however, has delocalized electrons spread across the three oxygen atoms. The delocalized electrons in ozone contribute to its unique reactivity and its ability to absorb UV radiation in the stratosphere.
3. Comparison of Conjugated Systems:
Conjugated systems, containing alternating single and multiple bonds, exhibit significant electron delocalization. The extent of delocalization depends on the length of the conjugated system. A longer conjugated system usually leads to greater delocalization and increased stability.
Delocalization and Molecular Orbital Theory:
Molecular orbital (MO) theory offers a more sophisticated understanding of electron delocalization. MO theory explains that electrons in a molecule occupy molecular orbitals that encompass the entire molecule, not just individual atoms or bonds. Delocalized electrons occupy bonding molecular orbitals that extend over several atoms, leading to the stabilization and characteristic properties discussed earlier.
Determining Delocalization:
Several factors influence the degree of electron delocalization:
- Conjugation: The presence of alternating single and multiple bonds promotes delocalization.
- Aromaticity: Aromatic compounds, possessing specific structural characteristics (e.g., Huckel's rule), exhibit extensive electron delocalization.
- Resonance: Multiple resonance structures contributing to the overall structure indicate delocalization.
- Molecular Orbital Theory: MO diagrams visually represent the extent of electron delocalization in molecular orbitals.
Conclusion:
The distinction between localized and delocalized electrons is fundamental to understanding molecular structure, bonding, and reactivity. Localized electrons are associated with specific atoms or bonds, resulting in predictable reactivity. Delocalized electrons, on the other hand, are spread over multiple atoms, leading to increased stability, unique reactivity patterns, and diverse properties impacting the macroscopic behavior of molecules and materials. Mastering this concept is essential for progressing in advanced chemistry and related disciplines. Further exploration into molecular orbital theory provides a deeper quantitative understanding of electron delocalization and its far-reaching consequences.
Latest Posts
Latest Posts
-
A Bundle Of Muscle Fibers Is Called
Apr 04, 2025
-
A Force Is A Scalar Quantity True False
Apr 04, 2025
-
A Sound Wave Is An Example Of A Longitudinal Wave
Apr 04, 2025
-
Fundamental Building Block Of All Matter
Apr 04, 2025
-
Exterior Angle Of A Regular Hexagon
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Difference Between Localized And Delocalized Electrons . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
