Difference Between Electron Pair Geometry And Molecular Geometry
Juapaving
Apr 02, 2025 · 6 min read
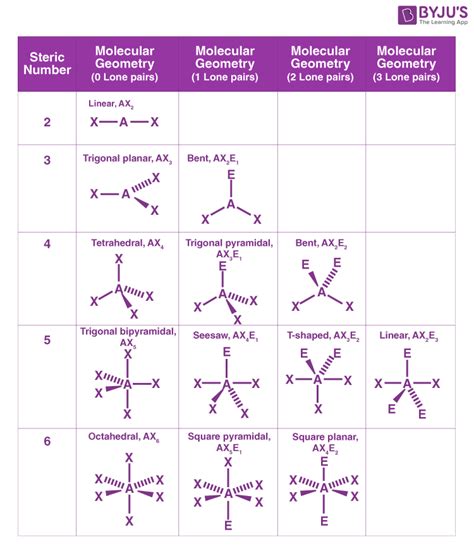
Table of Contents
Unveiling the Subtle Differences: Electron Pair Geometry vs. Molecular Geometry
Understanding the three-dimensional arrangement of atoms within a molecule is crucial in chemistry. This understanding forms the basis for predicting molecular properties like polarity, reactivity, and physical state. Two key concepts underpin this understanding: electron pair geometry and molecular geometry. While often used interchangeably, these terms represent distinct aspects of molecular structure, and grasping their differences is vital for mastering chemical concepts. This article delves into the intricacies of both, clarifying the distinctions and providing a framework for understanding their importance.
What is Electron Pair Geometry?
Electron pair geometry describes the arrangement of all electron pairs surrounding the central atom in a molecule. This includes both bonding pairs (electron pairs involved in covalent bonds) and lone pairs (electron pairs not involved in bonding). The electron pairs repel each other due to their negative charge, arranging themselves to minimize this repulsion and achieve maximum separation. This arrangement dictates the basic framework of the molecule's shape.
The Valence Shell Electron Pair Repulsion (VSEPR) theory is the foundational model used to predict electron pair geometry. VSEPR theory postulates that the electron pairs around a central atom will arrange themselves to be as far apart as possible to minimize electrostatic repulsion. This leads to several predictable electron pair geometries, each determined by the number of electron pairs surrounding the central atom:
Common Electron Pair Geometries:
- Linear: Two electron pairs are arranged 180° apart. Examples include BeCl₂ and CO₂.
- Trigonal Planar: Three electron pairs are arranged in a flat triangle with 120° angles between them. Examples include BF₃ and SO₃.
- Tetrahedral: Four electron pairs are arranged in a three-dimensional tetrahedron with 109.5° angles between them. Examples include CH₄ and SiCl₄.
- Trigonal Bipyramidal: Five electron pairs are arranged in a trigonal bipyramid. Three electron pairs lie in a plane forming a triangle (equatorial positions), and two electron pairs are located above and below this plane (axial positions). Examples include PCl₅ and SF₄.
- Octahedral: Six electron pairs are arranged in an octahedron with 90° angles between them. Examples include SF₆ and XeF₆.
What is Molecular Geometry?
Molecular geometry, also known as molecular shape, describes the three-dimensional arrangement of only the atoms in a molecule. Unlike electron pair geometry, it considers only the positions of the atoms, ignoring the lone pairs of electrons on the central atom. Lone pairs influence the molecular geometry by affecting the bond angles and overall shape. However, they are not explicitly part of the description of the molecular geometry itself.
The presence of lone pairs creates distortions in the ideal geometries predicted by VSEPR theory. The lone pairs exert a stronger repulsive force than bonding pairs, pushing the bonded atoms closer together. This results in deviations from the perfect symmetrical shapes associated with electron pair geometries.
Molecular Geometry Examples based on Electron Pair Geometry:
Let's illustrate the difference with some examples:
1. Methane (CH₄):
- Electron Pair Geometry: Tetrahedral (four bonding pairs)
- Molecular Geometry: Tetrahedral (four atoms arranged tetrahedrally)
In methane, since all four electron pairs are bonding pairs, the electron pair geometry and the molecular geometry are identical.
2. Ammonia (NH₃):
- Electron Pair Geometry: Tetrahedral (three bonding pairs and one lone pair)
- Molecular Geometry: Trigonal Pyramidal (three atoms arranged in a pyramid with the nitrogen atom at the apex)
The lone pair in ammonia repels the bonding pairs, causing a distortion from a perfect tetrahedron. The resulting molecular geometry is a trigonal pyramid.
3. Water (H₂O):
- Electron Pair Geometry: Tetrahedral (two bonding pairs and two lone pairs)
- Molecular Geometry: Bent (two atoms arranged with a bond angle less than 109.5°)
The two lone pairs in water significantly repel the bonding pairs, resulting in a bent molecular geometry with a bond angle of approximately 104.5°. This is considerably smaller than the ideal tetrahedral angle of 109.5°.
4. Carbon Dioxide (CO₂):
- Electron Pair Geometry: Linear (two bonding pairs)
- Molecular Geometry: Linear (two atoms arranged linearly)
Similar to methane, carbon dioxide has identical electron pair and molecular geometries because all electron pairs are bonding pairs.
5. Sulfur Hexafluoride (SF₆):
- Electron Pair Geometry: Octahedral (six bonding pairs)
- Molecular Geometry: Octahedral (six atoms arranged octahedrally)
Again, with only bonding pairs, electron pair and molecular geometries are the same.
The Importance of Distinguishing Between Electron Pair Geometry and Molecular Geometry
Understanding the difference between electron pair geometry and molecular geometry is crucial for several reasons:
-
Predicting Molecular Polarity: Molecular polarity, which affects a molecule's interaction with electric fields and other molecules, directly depends on the molecular geometry and the presence of polar bonds. Electron pair geometry alone is insufficient to determine polarity.
-
Determining Molecular Reactivity: The shape of a molecule dictates which parts are accessible for reactions. Molecular geometry, influenced by the presence of lone pairs, directly impacts reactivity.
-
Interpreting Spectroscopic Data: Spectroscopic techniques, such as infrared (IR) and nuclear magnetic resonance (NMR) spectroscopy, provide information about the molecular structure. Analysis of this data requires an understanding of both electron pair and molecular geometries.
-
Understanding Intermolecular Forces: The shapes of molecules determine the types and strengths of intermolecular forces (like hydrogen bonding, dipole-dipole interactions, and London dispersion forces), influencing physical properties such as boiling point and melting point.
Beyond the Basics: Advanced Considerations
While VSEPR theory provides a good starting point for predicting molecular geometries, more sophisticated computational methods are needed for complex molecules. These methods consider factors beyond simple electron-electron repulsion, including:
-
Hybridization: The concept of orbital hybridization explains the observed bond angles and shapes in many molecules. Hybrid orbitals are formed by mixing atomic orbitals, influencing the geometry.
-
Bond Order: Multiple bonds (double or triple bonds) exert a stronger repulsive force than single bonds, affecting the molecular geometry.
-
Steric Effects: The bulkiness of atoms and functional groups can also cause steric hindrance, influencing the observed molecular geometry.
-
Hyperconjugation: In certain molecules, the interaction between filled and empty orbitals can further affect the geometry.
Conclusion: A Unified Perspective
Electron pair geometry and molecular geometry are interconnected but distinct concepts. Electron pair geometry describes the arrangement of all electron pairs around the central atom, while molecular geometry focuses solely on the spatial arrangement of the atoms. Understanding the subtle yet significant difference between these two aspects is fundamental to comprehending molecular structure and its influence on various chemical and physical properties. By understanding both, you gain a comprehensive perspective on the three-dimensional world of molecules and their behavior. The use of VSEPR theory, combined with a deeper understanding of more advanced concepts, provides a powerful tool for predicting and explaining the behavior of molecules, advancing our understanding of the chemical world.
Latest Posts
Latest Posts
-
Kg M 3 To G Cm3
Apr 03, 2025
-
120 Cm Is How Many Inches
Apr 03, 2025
-
The Scattering Of Light By A Colloid Is Called The
Apr 03, 2025
-
Roots Forming A Number In Logo
Apr 03, 2025
-
Alloy Of Mercury With Another Metal
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Difference Between Electron Pair Geometry And Molecular Geometry . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
