Difference Between Atom And An Ion
Juapaving
Apr 02, 2025 · 6 min read
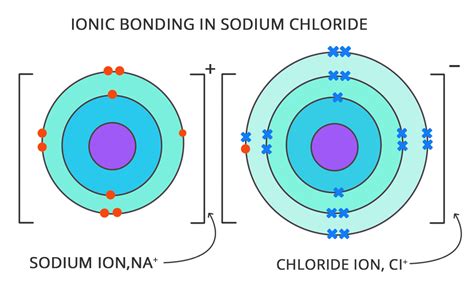
Table of Contents
Delving Deep: The Fundamental Difference Between Atoms and Ions
Understanding the building blocks of matter is crucial to grasping the complexities of chemistry and physics. At the heart of this understanding lies the difference between an atom and an ion. While seemingly subtle, this distinction is fundamental to how matter interacts and behaves. This article will delve deep into this difference, exploring their structures, properties, and the processes that transform one into the other. We will also touch upon their relevance in various fields, from material science to biology.
What is an Atom?
An atom is the basic unit of a chemical element. It's the smallest particle that retains the chemical properties of that element. Think of it as the fundamental building block of everything around us – from the air we breathe to the earth beneath our feet. Atoms are incredibly small; their size is measured in picometers (trillionths of a meter).
Structure of an Atom:
Atoms are composed of three fundamental subatomic particles:
- Protons: Positively charged particles found in the atom's nucleus (center). The number of protons determines the element's atomic number and its identity. For example, all hydrogen atoms have one proton, while all oxygen atoms have eight.
- Neutrons: Neutrally charged particles also located in the nucleus. They contribute to the atom's mass but not its charge. The number of neutrons can vary within the same element, resulting in isotopes. For example, carbon-12 and carbon-14 are isotopes of carbon, differing in the number of neutrons.
- Electrons: Negatively charged particles that orbit the nucleus in electron shells or energy levels. These shells have varying energy levels, with electrons in lower shells being closer to the nucleus and more tightly bound. The number of electrons typically equals the number of protons in a neutral atom, balancing the positive charge of the protons.
Atomic Number and Mass Number:
The atomic number uniquely identifies an element and is equal to the number of protons in its nucleus. The mass number represents the total number of protons and neutrons in the nucleus. Knowing these two numbers allows us to fully characterize an atom of a specific element.
What is an Ion?
An ion, on the other hand, is an atom or molecule that carries a net electrical charge. This charge arises from an imbalance in the number of protons and electrons. Unlike neutral atoms, ions are electrically charged and exhibit different properties compared to their neutral counterparts.
Formation of Ions:
Ions are formed through a process called ionization. This process involves the gain or loss of electrons from a neutral atom.
- Cation: When an atom loses one or more electrons, it becomes positively charged. This positively charged ion is called a cation. Metals tend to lose electrons and form cations easily. For example, a sodium atom (Na) can lose one electron to form a sodium cation (Na⁺).
- Anion: When an atom gains one or more electrons, it becomes negatively charged. This negatively charged ion is called an anion. Nonmetals tend to gain electrons and form anions readily. For example, a chlorine atom (Cl) can gain one electron to form a chloride anion (Cl⁻).
Properties of Ions:
The properties of ions differ significantly from their neutral atom counterparts:
- Charge: The most significant difference is the net electrical charge. Cations are positively charged, while anions are negatively charged.
- Reactivity: Ions are highly reactive due to their charge imbalance. They readily participate in chemical reactions, particularly ionic bonding, where oppositely charged ions attract each other to form stable compounds.
- Size: The size of an ion differs from that of its parent atom. Cations are generally smaller than their parent atoms because they have lost electrons, reducing electron-electron repulsion. Anions are generally larger than their parent atoms due to the added electrons increasing electron-electron repulsion.
- Physical Properties: Ions often exhibit different physical properties compared to their neutral atoms. This includes differences in melting and boiling points, solubility, and conductivity.
Key Differences Between Atoms and Ions:
The following table summarizes the key differences between atoms and ions:
| Feature | Atom | Ion |
|---|---|---|
| Charge | Neutral (no net charge) | Positive (cation) or negative (anion) |
| Electron Number | Equal to proton number | Unequal to proton number |
| Formation | Fundamental building block of matter | Formed by gain or loss of electrons |
| Reactivity | Relatively less reactive | Highly reactive |
| Size | Varies depending on the element | Varies, Cations smaller, Anions larger |
| Examples | Na, O, C | Na⁺, O²⁻, Cl⁻ |
Importance of Atoms and Ions:
Atoms and ions play critical roles in various fields:
Chemistry:
- Chemical bonding: Atoms and ions interact to form chemical bonds, which are the forces that hold atoms together in molecules and compounds. Ionic bonds are formed between oppositely charged ions, while covalent bonds involve the sharing of electrons between atoms.
- Chemical reactions: Chemical reactions involve the rearrangement of atoms and ions to form new substances. The reactivity of ions is crucial in many chemical processes.
- Stoichiometry: Stoichiometry is the quantitative study of reactants and products in chemical reactions. Understanding the number of atoms and ions involved is essential for accurate stoichiometric calculations.
Biology:
- Biological molecules: Biological molecules like proteins, carbohydrates, and nucleic acids are composed of atoms held together by chemical bonds.
- Electrolyte balance: Ions play a critical role in maintaining electrolyte balance in the body, which is essential for various physiological processes. Sodium (Na⁺), potassium (K⁺), and chloride (Cl⁻) ions are crucial examples.
- Enzyme function: Many enzymes require ions as cofactors to function properly. These ions help catalyze biochemical reactions.
- Nerve impulse transmission: The transmission of nerve impulses relies on the movement of ions across cell membranes.
Material Science:
- Crystal structures: The arrangement of ions in crystals determines many of the material's properties, such as hardness, conductivity, and melting point.
- Semiconductors: Semiconductors, materials used in electronics, often rely on the properties of ions and their ability to conduct electricity under specific conditions.
- Corrosion: Corrosion of metals involves the formation of metal ions, which are then oxidized, degrading the metal's structure.
Physics:
- Nuclear physics: The nucleus of an atom is the focus of nuclear physics, which explores nuclear reactions and radioactivity.
- Plasma physics: Plasma, the fourth state of matter, is composed of ions and free electrons. Plasma physics explores the behavior of plasmas and their applications.
Conclusion:
The difference between an atom and an ion is fundamental to understanding the structure and behavior of matter. While atoms are neutral and relatively unreactive, ions carry a net electrical charge and are highly reactive. This difference profoundly impacts their properties and their roles in various chemical, biological, material, and physical phenomena. A deep understanding of this distinction is crucial for anyone interested in exploring the intricacies of the natural world. Further exploration into the specifics of ionization processes and the various types of ions and their behaviors can lead to a more complete understanding of the fundamental nature of matter and energy. From the smallest subatomic particles to the largest structures in the universe, the concepts of atoms and ions provide a foundational understanding of the processes that shape our world.
Latest Posts
Latest Posts
-
Which Planets Do Not Have Moons
Apr 03, 2025
-
What Is The Perimeter Of A Parallelogram
Apr 03, 2025
-
Can A Pure Substance Be Separated By Physical Means
Apr 03, 2025
-
Does A Ribosome Have A Membrane
Apr 03, 2025
-
Is Aluminium A Conductor Of Electricity
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Difference Between Atom And An Ion . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
