Can Pure Substances Be Separated By Chemical Means
Juapaving
Apr 05, 2025 · 5 min read
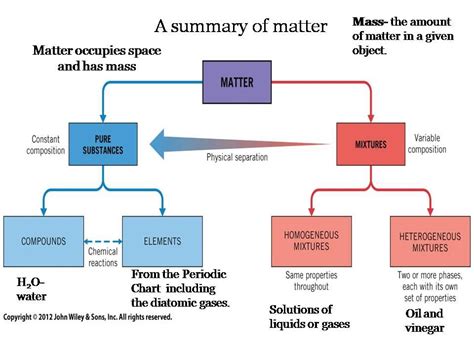
Table of Contents
Can Pure Substances Be Separated by Chemical Means?
The question of whether pure substances can be separated by chemical means is a nuanced one, demanding a deep understanding of both the nature of pure substances and the definition of chemical separation methods. The short answer is: yes, but not in the way you might initially think. Pure substances, by definition, are composed of only one type of atom or molecule. This seemingly simple definition leads to a more complex consideration of what constitutes a chemical separation.
Understanding Pure Substances
Before delving into separation techniques, let's establish a clear definition of a pure substance. A pure substance has a fixed chemical composition and uniform properties throughout. This means that no matter where you take a sample from a pure substance, its chemical makeup and physical characteristics will remain consistent. Examples include distilled water (H₂O), pure gold (Au), and table salt (NaCl). These are all examples of substances that have a defined chemical formula and predictable properties. Crucially, the properties of a pure substance are intrinsic and unchanging. They are not determined by the method of purification or the external factors applied.
It's vital to distinguish pure substances from mixtures. Mixtures consist of two or more substances that are physically combined, not chemically bonded. They can be separated into their constituent components by physical means, such as filtration, distillation, or chromatography. Unlike pure substances, the composition and properties of mixtures are not uniform; they can vary depending on the proportions of the components.
Chemical vs. Physical Separation Methods
The key to understanding the separation of pure substances lies in differentiating between chemical and physical methods. Physical separation techniques change the physical state or arrangement of substances without altering their chemical composition. Examples include:
- Filtration: Separating solids from liquids using a porous medium.
- Distillation: Separating liquids based on their boiling points.
- Evaporation: Separating a dissolved solid from a liquid by evaporating the solvent.
- Chromatography: Separating components of a mixture based on their differential affinities for a stationary and mobile phase.
- Centrifugation: Separating substances based on their density using centrifugal force.
Chemical separation methods, on the other hand, involve breaking and reforming chemical bonds, thus altering the chemical composition of the substances involved. These methods transform the chemical identity of the components, creating new substances. Examples include:
- Electrolysis: Using an electric current to decompose a compound into its constituent elements. For example, the electrolysis of water (H₂O) yields hydrogen (H₂) and oxygen (O₂).
- Chemical Reactions: Utilizing chemical reactions to selectively transform one component of a mixture into another, making it easier to separate. This can involve precipitation reactions, acid-base reactions, redox reactions, etc.
- Crystallization: A process often involving chemical reactions where a substance precipitates out of solution as a pure crystalline solid. This can be achieved by carefully manipulating the solution's conditions, such as temperature or pH.
Can a Pure Substance Be Separated by Chemical Means? A Deeper Dive
While a pure substance cannot be separated into different pure substances through physical means, it can be chemically altered into new substances. This is the crucial distinction. The chemical transformation creates different pure substances, but it's not a separation of the original pure substance into its constituents in the same way you'd separate components of a mixture.
Let's consider the example of water (H₂O) again. Water is a pure substance. Electrolysis breaks down water into hydrogen and oxygen gases. These are also pure substances. However, it's incorrect to say that electrolysis separated the water into its constituent elements in the same way you'd separate sand from water using filtration. Electrolysis is a chemical change; it fundamentally alters the chemical composition, creating new substances altogether.
Similarly, consider the decomposition of calcium carbonate (CaCO₃). When heated, calcium carbonate decomposes into calcium oxide (CaO) and carbon dioxide (CO₂). Both CaO and CO₂ are pure substances, but the process was a chemical change, not a separation of pre-existing components. The original pure substance (CaCO₃) no longer exists in its original form.
The Role of Chemical Reactions in "Separating" Pure Substances
Chemical reactions are often employed to isolate a specific element or compound from a mixture, even if it initially starts as a part of a pure substance. Let's say you have a pure sample of a complex organic molecule. Through a series of carefully controlled chemical reactions, you might selectively remove or modify certain functional groups on the molecule. This process results in the formation of one or more new pure substances, effectively "separating" them from the original molecule. It's a chemical transformation, not a separation in the classic sense.
Practical Applications: Refining and Synthesis
The concepts discussed here have significant implications in various fields. In metallurgy, for instance, the refining of metals often involves chemical processes. Crude ores, which are mixtures, undergo chemical treatments to extract the pure metal. This process isn't separating the metal from other metals within a pure substance; it's about separating the metal from impurities within a mixture.
In chemical synthesis, the goal is often to produce pure substances. Researchers and chemists employ a variety of chemical reactions to synthesize new compounds and isolate them from reaction byproducts. This process invariably involves chemical changes.
Conclusion: Redefining Separation
The question of whether pure substances can be separated by chemical means ultimately boils down to a matter of semantics. You cannot separate a pure substance into its constituent elements in the physical sense. However, you can chemically transform it into different pure substances. The act of creating new pure substances through chemical means could be interpreted as a form of separation if you define separation broadly enough to encompass chemical transformations.
It's vital to maintain a clear distinction between chemical and physical separation methods. While physical separation techniques deal with physical states and arrangements of substances, chemical separation methods involve altering the chemical composition. A pure substance can be chemically altered, leading to the production of new pure substances, but this is fundamentally different from separating the components of a mixture. Therefore, while the answer is "yes" in a broad sense, it's crucial to acknowledge the nuance and avoid misinterpreting the nature of chemical processes. The original pure substance ceases to exist upon chemical transformation, replaced by new, distinct pure substances.
Latest Posts
Latest Posts
-
What Percent Of 40 Is 32
Apr 06, 2025
-
What Is The Iupac Name For The Compound Shown
Apr 06, 2025
-
Is 29 A Composite Or Prime Number
Apr 06, 2025
-
Atomic Mass Equals The Number Of
Apr 06, 2025
-
Definition Of Democracy By Abraham Lincoln
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Can Pure Substances Be Separated By Chemical Means . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
