Ca Oh 2 Acid Or Base
Juapaving
Apr 03, 2025 · 5 min read
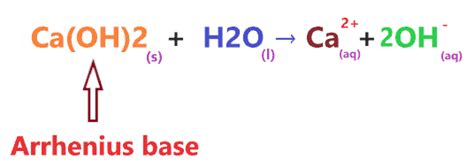
Table of Contents
Ca(OH)₂: A Deep Dive into its Acidic or Basic Nature
Calcium hydroxide, Ca(OH)₂, commonly known as slaked lime or hydrated lime, is a crucial inorganic compound with widespread applications across various industries. Understanding its chemical properties, particularly its acidic or basic nature, is fundamental to appreciating its diverse uses. This comprehensive article will explore the chemical behavior of Ca(OH)₂, clarifying its classification as a base and delving into the reasons behind this classification, its reactions, and its significant applications.
Understanding Acids and Bases
Before delving into the specifics of Ca(OH)₂, let's establish a clear understanding of acids and bases. Several theories exist to define acids and bases, but the most relevant for understanding Ca(OH)₂ are the Arrhenius and Brønsted-Lowry theories.
Arrhenius Theory
The Arrhenius theory defines acids as substances that produce hydrogen ions (H⁺) when dissolved in water, and bases as substances that produce hydroxide ions (OH⁻) when dissolved in water. This theory, while simple, is limited in its scope.
Brønsted-Lowry Theory
The Brønsted-Lowry theory offers a broader perspective. It defines acids as proton (H⁺) donors and bases as proton acceptors. This theory expands the definition beyond aqueous solutions, encompassing reactions in other solvents or even without a solvent.
Ca(OH)₂: A Strong Base
Ca(OH)₂ unequivocally falls into the category of a strong base. This classification stems from its behavior in aqueous solutions. When Ca(OH)₂ dissolves in water, it undergoes complete dissociation, releasing calcium ions (Ca²⁺) and hydroxide ions (OH⁻):
Ca(OH)₂(s) → Ca²⁺(aq) + 2OH⁻(aq)
The presence of a significant concentration of hydroxide ions (OH⁻) in the solution is the hallmark of a base. The higher the concentration of OH⁻ ions, the stronger the base. Ca(OH)₂'s complete dissociation ensures a high concentration of OH⁻, solidifying its status as a strong base. This complete dissociation is a key differentiator between strong and weak bases. Weak bases only partially dissociate, resulting in a lower concentration of OH⁻ ions.
pH and pOH: Quantifying Basicity
The basicity of a solution is quantifiable using the pH and pOH scales. The pH scale measures the concentration of hydrogen ions (H⁺), while the pOH scale measures the concentration of hydroxide ions (OH⁻). A pH of 7 indicates neutrality, while values below 7 indicate acidity and values above 7 indicate basicity. The pH and pOH are related by the equation:
pH + pOH = 14
Since Ca(OH)₂ produces a high concentration of OH⁻ ions, a solution containing Ca(OH)₂ will have a high pOH and consequently a high pH, confirming its basic nature.
Reactions of Ca(OH)₂
The strong basic nature of Ca(OH)₂ leads to a variety of characteristic reactions:
Neutralization Reactions
One of the most important reactions of Ca(OH)₂ is its neutralization reaction with acids. When Ca(OH)₂ reacts with an acid, it forms a salt and water. For example, the reaction with hydrochloric acid (HCl) is:
Ca(OH)₂(aq) + 2HCl(aq) → CaCl₂(aq) + 2H₂O(l)
This reaction is exothermic, meaning it releases heat. The heat generated can be significant, making it crucial to handle such reactions carefully. Similar neutralization reactions occur with other acids, like sulfuric acid (H₂SO₄) and nitric acid (HNO₃), forming the corresponding calcium salts.
Reaction with Carbon Dioxide
Ca(OH)₂ reacts with carbon dioxide (CO₂) to form calcium carbonate (CaCO₃) and water:
Ca(OH)₂(aq) + CO₂(g) → CaCO₃(s) + H₂O(l)
This reaction is used in various applications, including the removal of CO₂ from gases and the production of lime mortar in construction. The formation of the insoluble CaCO₃ is visually evident as a white precipitate.
Reaction with Metal Salts
Ca(OH)₂ can react with certain metal salts to form insoluble metal hydroxides. For instance, the reaction with copper(II) sulfate (CuSO₄) results in the formation of copper(II) hydroxide (Cu(OH)₂), a blue precipitate:
Ca(OH)₂(aq) + CuSO₄(aq) → CaSO₄(aq) + Cu(OH)₂(s)
This type of reaction is a precipitation reaction, often used in qualitative analysis for identifying the presence of certain metal ions.
Applications of Ca(OH)₂
The versatile properties of Ca(OH)₂ lead to its widespread applications in various fields:
Construction Industry
In the construction industry, Ca(OH)₂ serves as a key component in mortar, plaster, and whitewash. Its reaction with CO₂ in the air leads to the hardening of these materials, providing structural strength.
Water Treatment
Ca(OH)₂ plays a vital role in water treatment. It is used to adjust the pH of water, remove impurities, and soften hard water by precipitating out calcium and magnesium ions. It also aids in the removal of heavy metals from wastewater.
Chemical Industry
Ca(OH)₂ is used in various chemical processes as a base, neutralizing acids and participating in several chemical reactions. It serves as a precursor in the production of other calcium compounds.
Agriculture
In agriculture, Ca(OH)₂ is used to adjust soil pH, reducing soil acidity and making it more suitable for plant growth. It also provides calcium, an essential nutrient for plants.
Food Industry
In certain food applications, Ca(OH)₂ is used as a food additive. Its primary role is to control acidity, often in specific food processing methods and preparations. However, appropriate usage and compliance with regulations are critical.
Other Applications
Beyond these major applications, Ca(OH)₂ finds use in several other areas, including:
- Pulp and Paper Industry: Used in the pulping process and in the treatment of wastewater.
- Sugar Refining: Used in the purification of sugar.
- Leather Tanning: Employed in the tanning process to neutralize acids.
Safety Precautions
While Ca(OH)₂ has numerous applications, it's essential to handle it cautiously. Direct contact can cause skin and eye irritation. Inhalation of dust can irritate the respiratory tract. Therefore, appropriate safety measures, including the use of protective gear like gloves and eye protection, are crucial during handling.
Conclusion
Ca(OH)₂, definitively a strong base, showcases its multifaceted nature through its chemical behavior and diverse applications. From its role in construction to water treatment and various chemical processes, its impact across numerous industries is undeniable. Understanding its basic nature and its reactions is crucial for leveraging its potential while adhering to essential safety protocols. The versatility of Ca(OH)₂ continues to make it a cornerstone compound in various fields, highlighting its significance in modern applications. Further research into its properties and applications promises even wider applications in the future.
Latest Posts
Latest Posts
-
How Many Joules In A Kilowatt Hour
Apr 04, 2025
-
1 Nucleic Acids Are Polymers Of
Apr 04, 2025
-
How Many Centimeters Are In 2 M
Apr 04, 2025
-
What Is The Lowest Common Multiple Of 12 And 16
Apr 04, 2025
-
Factors That Affect The Rate Of Osmosis
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Ca Oh 2 Acid Or Base . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
