Benedict's Reagent Is Used To Identify Which Of The Following
Juapaving
Apr 05, 2025 · 5 min read
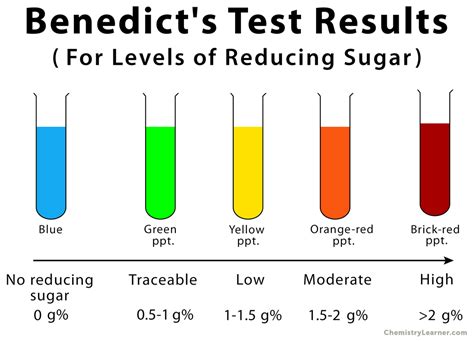
Table of Contents
Benedict's Reagent: A Comprehensive Guide to Identifying Reducing Sugars
Benedict's reagent is a widely used chemical reagent in analytical chemistry, primarily known for its ability to detect the presence of reducing sugars. Understanding its function, mechanism, and applications is crucial for various fields, from biochemistry and food science to clinical diagnostics. This comprehensive guide will delve into the intricacies of Benedict's reagent, explaining what it is used to identify, how it works, and its limitations.
What is Benedict's Reagent?
Benedict's reagent is a complex alkaline solution containing copper(II) sulfate, sodium citrate, and sodium carbonate. The copper(II) ions (Cu²⁺) are the key component responsible for the color change reaction. The sodium citrate acts as a complexing agent, preventing the precipitation of copper(II) hydroxide, while the sodium carbonate provides the alkaline environment necessary for the reaction to occur.
Key Components and their Roles:
- Copper(II) Sulfate (CuSO₄): Provides the copper(II) ions that undergo reduction in the presence of reducing sugars.
- Sodium Citrate (Na₃C₆H₅O₇): Prevents the formation of insoluble copper(II) hydroxide, ensuring the solution remains homogeneous.
- Sodium Carbonate (Na₂CO₃): Creates the alkaline environment required for the reaction to take place. The alkalinity maintains the copper ions in solution and facilitates the oxidation of the reducing sugar.
Identifying Reducing Sugars: The Core Application
The primary purpose of Benedict's reagent is to identify reducing sugars. Reducing sugars are carbohydrates that possess a free aldehyde (-CHO) or ketone (-C=O) group. This functional group can readily donate electrons, acting as a reducing agent. When a reducing sugar is present, it reduces the blue copper(II) ions (Cu²⁺) in Benedict's reagent to copper(I) ions (Cu⁺), which then precipitate as a reddish-brown copper(I) oxide (Cu₂O). The color change from blue to reddish-brown is indicative of a positive test for reducing sugars.
What Sugars are Detected?
Benedict's reagent can detect a wide range of reducing sugars, including:
- Glucose: A monosaccharide and the primary sugar in blood.
- Fructose: A monosaccharide found in fruits and honey.
- Galactose: A monosaccharide found in milk and other dairy products.
- Maltose: A disaccharide composed of two glucose units.
- Lactose: A disaccharide composed of glucose and galactose, found in milk.
Sugars NOT Detected by Benedict's Reagent
Importantly, Benedict's reagent does not detect all sugars. Non-reducing sugars, such as sucrose (table sugar), lack a free aldehyde or ketone group, and therefore cannot reduce the copper(II) ions. This is because sucrose's glycosidic linkage involves both the anomeric carbons, preventing the formation of a free aldehyde or ketone group needed for reduction.
The Mechanism of the Reaction
The reaction between Benedict's reagent and a reducing sugar is an oxidation-reduction (redox) reaction. The reducing sugar is oxidized, losing electrons, while the copper(II) ions are reduced, gaining electrons. The process can be summarized as follows:
-
Alkaline Conditions: The sodium carbonate in Benedict's reagent creates an alkaline environment. This environment is crucial because it facilitates the opening of the cyclic structure of reducing sugars, revealing the free aldehyde or ketone group.
-
Oxidation of Reducing Sugar: The free aldehyde or ketone group of the reducing sugar donates electrons to the copper(II) ions. This oxidation converts the reducing sugar into a carboxylic acid.
-
Reduction of Copper(II) Ions: The copper(II) ions (Cu²⁺) accept the electrons from the reducing sugar, undergoing reduction to copper(I) ions (Cu⁺).
-
Precipitation of Copper(I) Oxide: The copper(I) ions then react to form insoluble copper(I) oxide (Cu₂O), which precipitates out of the solution as a reddish-brown precipitate. The intensity of the color is directly proportional to the concentration of reducing sugar present.
Interpreting the Results: Color Changes and Concentration
The color change observed in the Benedict's test is crucial for interpreting the results:
- Blue: Indicates the absence of reducing sugars. The solution remains the original blue color of the copper(II) sulfate.
- Green: Indicates a low concentration of reducing sugars.
- Yellow: Indicates a moderate concentration of reducing sugars.
- Orange: Indicates a high concentration of reducing sugars.
- Reddish-brown/Brick-red: Indicates a very high concentration of reducing sugars.
It is important to note that the intensity of the color change is not always a precise quantitative measurement. For accurate quantification of reducing sugars, other methods like spectrophotometry are typically employed.
Applications of Benedict's Test
Benedict's test finds wide applications in various fields:
-
Clinical Diagnostics: Detecting glucose in urine is a crucial diagnostic tool for monitoring diabetes mellitus. The presence of glucose in urine (glycosuria) indicates high blood glucose levels. Although less precise than blood glucose meters, it serves as a screening tool.
-
Food Science: Identifying and quantifying reducing sugars in food products such as fruits, honey, and dairy products. This is valuable in quality control and nutritional analysis.
-
Biochemistry: Identifying and characterizing reducing sugars in biological samples, playing a role in carbohydrate metabolism studies and research.
Limitations of Benedict's Test
While Benedict's test is a simple and useful method, it has some limitations:
-
Lack of Specificity: It does not identify the specific type of reducing sugar present. Only the presence or absence, and relative concentration, can be determined.
-
Interfering Substances: Certain substances can interfere with the test, leading to false positive or false negative results.
-
Sensitivity: It is not as sensitive as some modern techniques for detecting low concentrations of reducing sugars. For very low concentrations, other more sensitive methods might be necessary.
-
Qualitative vs. Quantitative: Primarily a qualitative test, indicating the presence or absence, and a rough estimate of the concentration, rather than providing an exact quantitative measurement.
Conclusion: A Valuable Tool in the Chemist's Arsenal
Benedict's reagent remains a valuable and widely used tool in detecting reducing sugars. Its simplicity, low cost, and readily available components make it accessible for various applications. Understanding its mechanism, limitations, and interpretation of results is essential for accurate and meaningful analyses. While it may not provide the precision of more sophisticated techniques, Benedict's test serves as a fundamental and practical method for identifying the presence of these important carbohydrates in a range of samples. Its continued use underscores its significance as a cornerstone technique in chemistry and related fields. Remembering its limitations, and employing it appropriately alongside other analytical methods, maximizes its effectiveness and provides a valuable insight into the presence and concentration of reducing sugars in the sample being analyzed.
Latest Posts
Latest Posts
-
How Many Litres In Cubic Foot
Apr 05, 2025
-
50 Yards Is How Many Feet
Apr 05, 2025
-
Bronsted Lowry Vs Lewis Vs Arrhenius
Apr 05, 2025
-
Name Two Constituents Of Baking Powder
Apr 05, 2025
-
The Study Of Tissue Is Known As
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Benedict's Reagent Is Used To Identify Which Of The Following . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
