An Element In The Nitrogen Family
Juapaving
Apr 05, 2025 · 6 min read
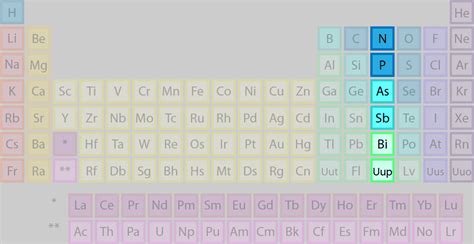
Table of Contents
Delving Deep into Phosphorus: An Element of the Nitrogen Family
Phosphorus, a fascinating element residing in Group 15 (also known as the nitrogen family) of the periodic table, plays a crucial role in numerous biological and industrial processes. Unlike its lighter congener nitrogen, which exists predominantly as a diatomic gas, phosphorus is a solid at room temperature and exhibits a far greater diversity in its allotropic forms and chemical reactivity. This article will delve deep into the properties, occurrence, extraction, uses, and biological significance of phosphorus, highlighting its unique characteristics within the nitrogen family.
The Unique Chemistry of Phosphorus: A Comparison with Nitrogen
Both phosphorus and nitrogen share the same valence electron configuration, leading to some similarities in their chemical behavior. However, the differences are striking, primarily due to the differences in their atomic sizes and electronegativity. Nitrogen, being smaller and more electronegative, forms stronger bonds and is more likely to form triple bonds, resulting in its stable diatomic N₂ molecule. Phosphorus, being larger and less electronegative, prefers to form single bonds and exhibits a greater tendency for catenation (the ability to form chains of atoms).
Key Differences:
- Allotropes: Nitrogen exists primarily as N₂, while phosphorus exhibits various allotropes, the most common being white phosphorus (highly reactive and toxic), red phosphorus (less reactive), and black phosphorus (least reactive). This allotropic variation significantly influences its reactivity and applications.
- Bonding: Nitrogen readily forms triple bonds, while phosphorus prefers single bonds. This difference leads to contrasting chemical behavior and stability.
- Oxidation States: Both elements exhibit a wide range of oxidation states, but phosphorus displays a greater preference for positive oxidation states in many of its compounds.
- Reactivity: White phosphorus is exceptionally reactive, igniting spontaneously in air, while red phosphorus is significantly less reactive. Nitrogen gas, on the other hand, is remarkably inert.
Occurrence and Extraction of Phosphorus
Phosphorus is not found free in nature due to its high reactivity. Instead, it occurs abundantly in various phosphate minerals, the most commercially important being apatite, a calcium phosphate mineral with the general formula Ca₅(PO₄)₃(OH,Cl,F). Vast deposits of apatite are found worldwide, serving as the primary source of phosphorus for industrial applications.
The Extraction Process:
The extraction of phosphorus from phosphate rock involves a high-temperature process in an electric arc furnace. The phosphate rock is first treated with sulfuric acid to produce phosphoric acid, which is then reduced with coke (carbon) and silica (SiO₂) at temperatures exceeding 1400°C. This process yields elemental phosphorus as a vapor, which is then condensed to a liquid and subsequently purified.
The overall reaction can be simplified as follows:
2Ca₃(PO₄)₂ + 6SiO₂ + 10C → 6CaSiO₃ + 10CO + P₄
This process, while effective, is energy-intensive and contributes to greenhouse gas emissions. Therefore, research is ongoing to develop more sustainable and environmentally friendly methods for phosphorus extraction.
The Diverse Applications of Phosphorus
Phosphorus, in its various forms and compounds, finds extensive applications across diverse sectors:
Fertilizers:
The overwhelming majority of phosphorus production is used in the production of phosphate fertilizers. Phosphorus is an essential nutrient for plant growth, promoting root development and flowering. The most common phosphate fertilizers include diammonium phosphate ((NH₄)₂HPO₄), monoammonium phosphate (NH₄H₂PO₄), and triple superphosphate (Ca(H₂PO₄)₂·H₂O). The global demand for phosphate fertilizers is immense, driven by the ever-increasing need to feed the growing world population. Concerns about the sustainability of phosphorus resources are growing, highlighting the importance of responsible fertilizer management and the exploration of alternative nutrient sources.
Detergents:
Phosphates were once widely used as water softeners in detergents. However, their use has been significantly reduced in many countries due to environmental concerns. Excessive phosphate runoff into water bodies can lead to eutrophication, causing algal blooms and oxygen depletion, harming aquatic life. Alternatives to phosphate-based detergents are now widely used.
Metallurgy:
Phosphorus is employed in metallurgy as a deoxidizer and alloying agent in the production of various metals, including steel and bronze. It can improve the strength and castability of these metals.
Matches:
Red phosphorus was historically a crucial component in match heads, providing the ignitable material. While safety matches are now widely used, red phosphorus still has applications in some specialized ignitable materials.
Other Applications:
Phosphorus compounds find applications in numerous other areas, including:
- Pesticides: Certain organophosphorus compounds are used as insecticides and herbicides.
- Flame Retardants: Organophosphorus compounds are employed as flame retardants in various materials, improving their fire safety.
- Food Additives: Phosphates are used as food additives to enhance texture, preserve quality, and prevent discoloration.
- Pharmaceuticals: Phosphorus plays a critical role in numerous pharmaceutical products.
The Biological Importance of Phosphorus
Phosphorus is an essential element for all living organisms. It is a vital component of:
- DNA and RNA: Phosphorus is present in the backbone of DNA and RNA molecules, the genetic material of all living things. The phosphodiester bonds linking nucleotides are crucial for the structure and function of nucleic acids.
- ATP: Adenosine triphosphate (ATP) is the primary energy currency of cells. The high-energy phosphate bonds in ATP provide the energy for cellular processes.
- Phospholipids: Phospholipids are major components of cell membranes, forming the lipid bilayer that separates the cell's interior from its surroundings.
- Bones and Teeth: In vertebrates, phosphorus is a key component of bones and teeth, contributing to their structural integrity.
The crucial role of phosphorus in biological systems highlights the importance of maintaining adequate phosphorus levels in the environment and ensuring sustainable phosphorus management practices. The depletion of phosphorus resources poses a significant challenge for agriculture and food security.
Environmental Concerns Related to Phosphorus
The extensive use of phosphorus in agriculture and industry has raised significant environmental concerns:
- Eutrophication: Phosphate runoff from agricultural fields and wastewater treatment plants contributes to eutrophication in water bodies. This process leads to excessive algal growth, oxygen depletion, and the death of aquatic life.
- Phosphorus Mining and its Impact: The mining of phosphate rock can have adverse environmental effects, including habitat destruction, soil erosion, and water pollution.
- Wastewater Treatment: Effective wastewater treatment is crucial to prevent phosphorus from entering waterways and causing environmental damage.
- Sustainable Phosphorus Management: Developing sustainable phosphorus management strategies is essential to ensure long-term phosphorus availability and minimize environmental impacts.
The Future of Phosphorus: Research and Development
Research is underway to address the challenges related to phosphorus availability and environmental impact:
- Improved Fertilizer Management: Developing strategies for more efficient fertilizer use can reduce phosphorus runoff and minimize environmental pollution.
- Phosphate Recycling: Recycling phosphorus from wastewater and other sources can help to conserve this valuable resource.
- Alternative Phosphorus Sources: Exploring alternative sources of phosphorus, such as recycled phosphorus from sewage sludge or struvite recovery, can reduce our dependence on mined phosphate rock.
- Developing Sustainable Agricultural Practices: Promoting sustainable agricultural practices, such as crop rotation and no-till farming, can help to minimize phosphorus loss from agricultural soils.
Conclusion: A Vital Element with Global Significance
Phosphorus, despite being less prominent in everyday conversation than other elements, is a cornerstone of life and a critical component of modern society. Its unique chemistry, diverse applications, and vital biological role make it an element worthy of in-depth study. Addressing the environmental challenges associated with phosphorus use and promoting sustainable management practices are crucial to ensuring the long-term availability of this vital resource for future generations. The ongoing research and development in this field are vital to securing a sustainable future and mitigating the environmental impact of phosphorus usage while ensuring its continued contribution to human well-being and agricultural productivity. The future of phosphorus management demands a multi-faceted approach, integrating scientific advancements with policy changes and responsible practices across industries.
Latest Posts
Latest Posts
-
The Most Reactive Group Of The Nonmetals Are The
Apr 06, 2025
-
Venn Diagram Cellular Respiration And Photosynthesis
Apr 06, 2025
-
What Joint Do You Use To Lift The Weight
Apr 06, 2025
-
Balanced Chemical Equation For Neutralization Of Acid With Naoh
Apr 06, 2025
-
Why Is Ice Melting Not A Chemical Reaction
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about An Element In The Nitrogen Family . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
