A Solution With A Ph Of 6 Is
Juapaving
Apr 05, 2025 · 5 min read
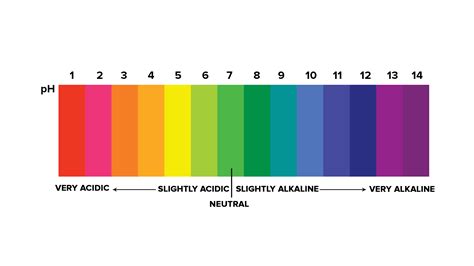
Table of Contents
A Solution with a pH of 6: Understanding Acidity, Applications, and Safety
A solution with a pH of 6 is considered slightly acidic. This seemingly simple statement opens a door to a vast world of chemical properties, practical applications, and safety considerations. Understanding the implications of a pH 6 solution is crucial across various fields, from environmental science and agriculture to industrial processes and human health. This article delves deep into the characteristics of pH 6 solutions, exploring their properties, common examples, and the importance of handling them safely.
Understanding the pH Scale
Before diving into the specifics of a pH 6 solution, it's essential to grasp the fundamentals of the pH scale. The pH scale measures the acidity or alkalinity of a solution, ranging from 0 to 14. A pH of 7 is considered neutral, representing pure water. Solutions with a pH below 7 are acidic, while those above 7 are alkaline (or basic). The scale is logarithmic, meaning each whole number change represents a tenfold change in acidity or alkalinity. For instance, a solution with a pH of 5 is ten times more acidic than a solution with a pH of 6.
The Significance of pH 6
A pH of 6 sits firmly in the slightly acidic range. While not intensely acidic like solutions with pH values closer to 0 (like battery acid), it’s still acidic enough to have noticeable effects on various substances and processes. The slight acidity can influence chemical reactions, affect biological systems, and even impact materials.
Common Examples of pH 6 Solutions
Many naturally occurring and man-made substances exhibit a pH around 6. Understanding these examples helps contextualize the implications of this pH level:
1. Natural Sources:
- Slightly acidic rainwater: While pure water has a neutral pH of 7, rainwater often absorbs carbon dioxide from the atmosphere, forming carbonic acid, resulting in a slightly acidic pH, often around 5.6 but can range up to 6 depending on various atmospheric conditions.
- Some fruits and vegetables: Certain fruits like apples and grapes, and some vegetables like tomatoes, possess a slightly acidic pH in their juices. This acidity contributes to their taste and preservation properties.
- Soil: Soil pH varies greatly depending on its composition and location. However, many soils exhibit a pH range between 5.5 and 6.5, falling within the slightly acidic range. This soil acidity influences nutrient availability to plants, affecting plant growth and health.
2. Manufactured Solutions:
- Some food preservatives: Some food preservatives are designed to maintain a slightly acidic environment to inhibit the growth of microorganisms and extend shelf life.
- Certain cleaning solutions: Some cleaning solutions, particularly those used for specific purposes like descaling or cleaning certain surfaces, might have a pH of around 6. However, it’s crucial to always check the product label for accurate pH information.
- Industrial processes: Several industrial processes utilize solutions with a pH of 6 for specific chemical reactions or material treatments. Examples could include certain etching processes or water treatment procedures.
Impacts and Applications of pH 6 Solutions
The slightly acidic nature of a pH 6 solution influences its interactions with various substances and systems:
1. Chemical Reactions:
The acidity of a pH 6 solution can catalyze or inhibit specific chemical reactions. This property is utilized in various chemical processes, including some types of synthesis, catalysis, and material modification. The specific reactions are highly dependent on the other chemicals present in the solution and the reaction conditions.
2. Biological Effects:
A pH 6 environment can significantly affect biological systems. While not as harsh as stronger acids, the slightly acidic conditions can influence the activity of enzymes, the growth of microorganisms, and the overall functioning of biological processes. For instance, it can impact the growth of certain bacteria and fungi in a given environment. This factor is crucial in areas such as food preservation, agriculture, and microbiology.
3. Material Interactions:
The slightly acidic nature of a pH 6 solution can affect the properties of certain materials. It may cause corrosion or degradation of specific metals or erode certain types of stone or concrete over time. However, the effect greatly depends on the material's composition and its resistance to acidic environments. This understanding is key to material selection and preservation in diverse applications.
Safety Precautions when Handling pH 6 Solutions
While a pH 6 solution is not as hazardous as strongly acidic solutions, it’s still important to exercise caution:
- Eye protection: Always wear appropriate eye protection, such as safety goggles, when handling any solution. Accidental splashes can irritate or damage the eyes.
- Protective gloves: Depending on the specific solution and the duration of handling, protective gloves may be necessary to prevent skin irritation or chemical burns. Different glove materials offer varying levels of protection against different chemicals, so the selection depends on the specific solution composition.
- Appropriate ventilation: Ensure adequate ventilation when working with any solution, especially in enclosed spaces. Certain solutions may release volatile compounds that can be harmful if inhaled.
- Proper disposal: Dispose of pH 6 solutions according to local regulations and guidelines. Never pour them down the drain without checking for specific disposal procedures for chemical wastes.
- Labeling: Clearly label containers holding pH 6 solutions to avoid accidental ingestion or misidentification. The label should contain information about the contents, concentration, and any specific hazards.
Conclusion
A solution with a pH of 6, while seemingly innocuous, holds significant implications across various scientific and practical domains. Its slightly acidic nature influences chemical reactions, impacts biological systems, affects the properties of materials, and necessitates appropriate safety measures during handling. Understanding the properties and implications of pH 6 solutions is essential for researchers, industrial workers, agricultural practitioners, and anyone working with chemicals in any capacity. This knowledge is critical for ensuring safety, maximizing efficiency, and minimizing any potential negative consequences. Remember to always consult safety data sheets (SDS) for specific information concerning the handling, storage, and disposal of any solution. While this article provides general guidance, the specific precautions may differ depending on the exact composition and concentration of the solution.
Latest Posts
Latest Posts
-
Purines Are Smaller Or Larger Than Pyrimidines
Apr 05, 2025
-
Which Enzyme Is Responsible For Adding Nucleotides
Apr 05, 2025
-
Describing Words That Start With W
Apr 05, 2025
-
Puberty Refers To A Period Of
Apr 05, 2025
-
What Is The Least Common Multiple Of 12 And 20
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about A Solution With A Ph Of 6 Is . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
