Purines Are Smaller Or Larger Than Pyrimidines
Juapaving
Apr 05, 2025 · 6 min read
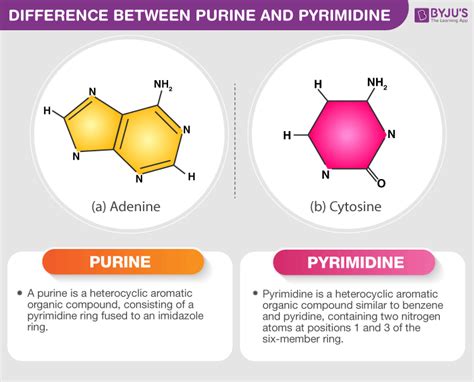
Table of Contents
Purines vs. Pyrimidines: Unveiling the Size Difference and its Significance
Understanding the fundamental building blocks of nucleic acids – DNA and RNA – is crucial in comprehending the intricate mechanisms of life. These building blocks are nucleotides, each composed of a nitrogenous base, a pentose sugar (ribose or deoxyribose), and a phosphate group. The nitrogenous bases are further categorized into two families: purines and pyrimidines. While both are crucial for the structure and function of DNA and RNA, a key difference lies in their size. This article delves into the comparative size of purines and pyrimidines, exploring the structural basis for this difference and its profound implications for DNA structure, stability, and function.
The Structural Basis of Size Difference: A Detailed Comparison
The core distinction between purines and pyrimidines lies in their chemical structure, directly influencing their size. This structural difference ultimately impacts their interactions within the DNA double helix and RNA structures.
Purines: The Larger Nitrogenous Bases
Purines are characterized by a double-ring structure. This structure consists of a six-membered ring fused to a five-membered ring, both containing nitrogen atoms. The two most common purines found in DNA and RNA are adenine (A) and guanine (G). Their larger, more complex structure contributes to their greater molecular weight and overall size compared to pyrimidines.
Adenine (A): This purine base possesses an amino group (-NH2) at the 6-position and a double bond at the 7-8 position. This specific arrangement of functional groups contributes to its hydrogen bonding capabilities, crucial for base pairing.
Guanine (G): Guanine contains a carbonyl group (=O) at the 6-position and an amino group (-NH2) at the 2-position. The presence of both a carbonyl and an amino group influences its hydrogen bonding properties and contributes to its interactions within the DNA double helix.
Pyrimidines: The Smaller Nitrogenous Bases
In contrast to purines, pyrimidines feature a single six-membered ring structure containing nitrogen atoms. The three main pyrimidine bases found in DNA and RNA are cytosine (C), thymine (T), and uracil (U). Their simpler, single-ring structure results in a smaller molecular weight and overall size compared to purines.
Cytosine (C): Cytosine possesses an amino group (-NH2) at the 4-position and a carbonyl group (=O) at the 2-position. These functional groups are essential for hydrogen bonding with guanine.
Thymine (T): Found exclusively in DNA, thymine contains two carbonyl groups (=O) at the 2- and 4-positions and a methyl group (-CH3) at the 5-position. The methyl group distinguishes thymine from uracil.
Uracil (U): Found only in RNA, uracil is similar to thymine, but lacks the methyl group at the 5-position. This subtle structural difference plays a role in the functional distinctions between DNA and RNA.
The Significance of Size Difference in DNA Structure and Function
The size difference between purines and pyrimidines is not merely a structural curiosity; it has profound implications for the very architecture and function of DNA.
Maintaining the DNA Double Helix: Chargaff's Rules and Base Pairing
The precise arrangement of purines and pyrimidines within the DNA double helix is dictated by Chargaff's rules, which state that the amount of adenine (A) always equals the amount of thymine (T), and the amount of guanine (G) always equals the amount of cytosine (C). This is because purines (A and G) always pair with pyrimidines (T and C respectively) through hydrogen bonds.
The pairing of a purine with a pyrimidine is crucial for maintaining the consistent diameter of the DNA double helix. If two purines were to pair, the resulting structure would bulge outwards, while two pyrimidines would cause a constriction. The purine-pyrimidine pairing ensures a uniform width, contributing to the overall stability and integrity of the DNA molecule.
Hydrogen Bonding: Specificity and Stability
The hydrogen bonds formed between base pairs (A-T and G-C) are fundamental for the stability of the DNA double helix. The specific number of hydrogen bonds differs between base pairs: A-T pairs have two hydrogen bonds, while G-C pairs have three. This difference in bonding strength influences the stability of different regions of the DNA molecule.
Implications for DNA Replication and Transcription
The precise base pairing between purines and pyrimidines is essential for accurate DNA replication and transcription. During replication, the DNA molecule unwinds, and each strand serves as a template for the synthesis of a new complementary strand. The specific pairing ensures that the newly synthesized strands are identical to the original strands, maintaining the genetic information. Similarly, during transcription, the DNA sequence is transcribed into RNA, with base pairing playing a critical role in ensuring accurate RNA synthesis.
Size and the DNA Groove Structure
The differential size of purines and pyrimidines contributes to the formation of the major and minor grooves in the DNA double helix. These grooves provide access points for proteins that interact with DNA, such as transcription factors and DNA-binding proteins. The specific dimensions of the grooves are influenced by the sizes and shapes of the purine and pyrimidine bases.
Beyond DNA: Purines and Pyrimidines in RNA
While the double-helical structure of DNA is iconic, RNA adopts more diverse conformations. Although the same purine and pyrimidine bases are found in RNA (A, G, C, and U), the absence of thymine and the presence of uracil contribute to RNA's unique structural features.
RNA Secondary Structures: Stem-Loops and Hairpins
RNA's single-stranded nature allows it to form complex secondary structures, including stem-loops and hairpins. These structures are stabilized by hydrogen bonding between complementary base pairs (A-U and G-C). The size difference between purines and pyrimidines again plays a role in the formation and stability of these secondary structures.
RNA's Functional Diversity
RNA molecules exhibit a wide range of functions, including acting as messenger molecules (mRNA), transferring amino acids during protein synthesis (tRNA), and forming structural components of ribosomes (rRNA). The specific structure and function of each RNA molecule is influenced by the sequence and arrangement of its purine and pyrimidine bases.
Purines and Pyrimidines in Metabolism and Disease
Purines and pyrimidines are not merely structural components; they are also actively involved in metabolic pathways. Disruptions in these metabolic pathways can lead to a variety of diseases.
Purine Metabolism and Gout
Gout is a painful inflammatory disease caused by the accumulation of uric acid crystals in the joints. Uric acid is the end product of purine metabolism, and elevated levels can lead to gout.
Pyrimidine Metabolism and Cancer
Disruptions in pyrimidine metabolism have been implicated in the development of certain cancers. Deficiencies or imbalances in pyrimidine biosynthesis can affect DNA replication and repair, potentially leading to genomic instability and tumor formation.
Conclusion: The Vital Role of Size and Structure
The seemingly minor difference in size between purines and pyrimidines has far-reaching consequences for the structure, stability, and function of DNA and RNA. This size difference dictates base pairing specificity, influences DNA double helix dimensions, and contributes to the diverse conformations adopted by RNA molecules. Understanding this fundamental difference is key to appreciating the intricate molecular mechanisms underlying life processes and the implications of metabolic dysregulation in disease. Further research into the nuances of purine and pyrimidine interactions promises to unveil even more profound insights into the molecular basis of life.
Latest Posts
Latest Posts
-
Is Iodine A Metal Or Nonmetal
Apr 06, 2025
-
What Is The Least Common Factor Of 5 And 7
Apr 06, 2025
-
The Most Reactive Metals Are The
Apr 06, 2025
-
How Many 1000 Are In A Million
Apr 06, 2025
-
5 Letter Word E N D
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Purines Are Smaller Or Larger Than Pyrimidines . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
