A Red Blood Cell Placed In A Hypertonic Medium Will
Juapaving
Apr 02, 2025 · 6 min read
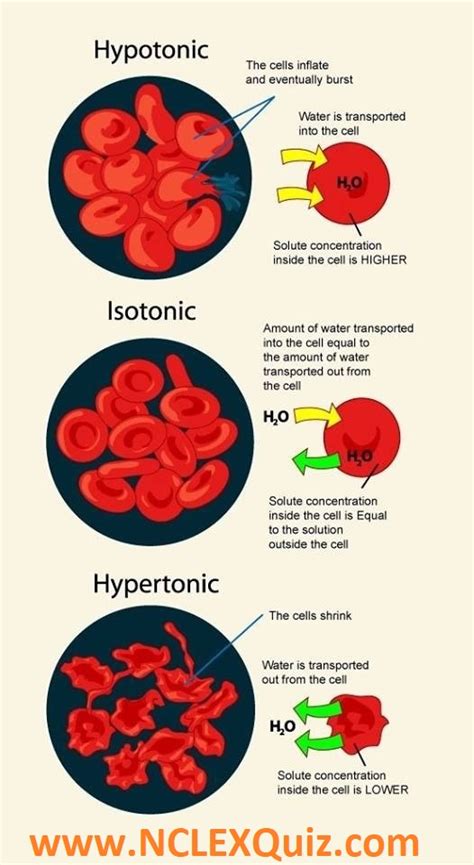
Table of Contents
A Red Blood Cell Placed in a Hypertonic Medium Will: Understanding Osmosis and its Effects
Red blood cells, the essential oxygen carriers in our bloodstream, are incredibly sensitive to their surrounding environment. Their behavior, particularly their shape and function, is heavily influenced by the tonicity of the solution they are immersed in. Understanding what happens to a red blood cell (RBC) placed in a hypertonic medium is crucial for comprehending fundamental biological principles and various medical conditions. This article will delve deep into this fascinating topic, exploring the process of osmosis, the effects of hypertonic solutions on RBCs, and the broader implications of this cellular phenomenon.
Understanding Osmosis: The Movement of Water Across Membranes
Before we dive into the specifics of red blood cells in hypertonic solutions, it's essential to grasp the concept of osmosis. Osmosis is the passive movement of water molecules across a selectively permeable membrane from a region of high water concentration to a region of low water concentration. This movement continues until equilibrium is reached, meaning the water concentration is equal on both sides of the membrane. The driving force behind osmosis is the difference in water potential, which is influenced by the concentration of solutes (dissolved substances) in the solution.
Selectively Permeable Membranes: The Gatekeepers of the Cell
The cell membrane of a red blood cell is a selectively permeable membrane. This means it allows certain molecules to pass through freely while restricting the passage of others. Water molecules can pass relatively easily through the membrane via aquaporins (water channels), but larger molecules and charged ions require specialized transport mechanisms. This selective permeability is crucial for maintaining the cell's internal environment and its ability to function properly.
Hypertonic Solutions: A High Concentration of Solutes
A hypertonic solution is one that has a higher solute concentration compared to the solution it's being compared to – in this case, the intracellular fluid of the red blood cell. This means there is a relatively lower concentration of water outside the cell compared to inside. When a red blood cell is placed in a hypertonic solution, the water potential gradient drives the movement of water out of the cell and into the surrounding solution.
What Happens to a Red Blood Cell in a Hypertonic Solution? Crenation
The movement of water out of the red blood cell due to the hypertonic environment leads to a process called crenation. Crenation is the shrinking of the red blood cell as it loses water. The cell membrane becomes wrinkled and distorted, losing its characteristic biconcave shape. The extent of crenation depends on the concentration of the hypertonic solution and the duration of exposure. A mildly hypertonic solution might cause slight shrinkage, while a highly concentrated hypertonic solution can lead to severe crenation and potentially cell death.
The Mechanism of Crenation: A Step-by-Step Look
- Water Potential Gradient: A hypertonic solution has a lower water potential than the cytoplasm of the red blood cell.
- Water Movement: Water molecules move passively across the cell membrane from the higher water potential (inside the cell) to the lower water potential (outside the cell).
- Cell Shrinkage: The loss of water causes the cell's volume to decrease, leading to crenation.
- Membrane Distortion: The cell membrane becomes wrinkled and folded due to the reduction in cell volume.
- Potential Cell Damage: Severe crenation can damage the cell membrane, potentially leading to cell lysis (rupture) in extreme cases or impaired cellular function.
The Importance of Maintaining Isotonic Conditions for RBC Function
Maintaining the isotonic state—where the solute concentration is equal inside and outside the cell—is vital for the proper functioning of red blood cells. In an isotonic environment, there is no net movement of water, and the cells maintain their shape and volume. This allows them to effectively carry oxygen throughout the body. Deviations from isotonicity, such as placing red blood cells in hypertonic or hypotonic solutions, disrupt their normal function and can lead to serious consequences.
Hypotonic Solutions: The Opposite Extreme
In contrast to hypertonic solutions, hypotonic solutions have a lower solute concentration than the intracellular fluid of the red blood cell. This results in a net movement of water into the cell, causing it to swell. In extreme cases, the cell can lyse (burst) due to the excessive influx of water. This process is called hemolysis.
Real-World Applications and Medical Implications
Understanding the effects of hypertonic solutions on red blood cells has significant implications in various medical fields:
1. Intravenous Fluid Administration: Maintaining Fluid Balance
The concentration of intravenous (IV) fluids is carefully controlled to avoid disrupting the tonicity of the blood. Using hypertonic solutions intravenously can cause dehydration and crenation of red blood cells, potentially leading to circulatory problems and organ damage. Isotonic solutions are typically used to maintain proper hydration and electrolyte balance.
2. Dehydration: Understanding the Cellular Consequences
Dehydration, a condition characterized by a significant loss of bodily fluids, can lead to a hypertonic environment in the bloodstream. The lack of water causes the blood to become more concentrated, resulting in crenation of red blood cells and impaired oxygen transport. Severe dehydration can have serious consequences for overall health.
3. Osmotic Diuretics: Utilizing Osmosis for Therapeutic Benefit
Some diuretics, medications used to increase urine production, work by creating an osmotic gradient in the kidneys. These osmotic diuretics draw water from the body into the urine, helping to reduce fluid retention. However, improper use can lead to imbalances and dehydration.
4. Preservation of Blood Samples: Maintaining Cellular Integrity
In laboratory settings, the preservation of blood samples requires careful consideration of tonicity. Using inappropriate solutions during sample collection and storage can lead to crenation or hemolysis, compromising the accuracy of blood tests and analyses.
5. Understanding Certain Pathological Conditions
Certain pathological conditions can alter the tonicity of body fluids, leading to abnormal red blood cell morphology and function. These conditions need to be addressed to restore fluid balance and cellular health.
Conclusion: The Delicate Balance of Tonicity
The behavior of a red blood cell placed in a hypertonic medium highlights the delicate balance that cells must maintain to function correctly. The process of osmosis and the consequences of altered tonicity are fundamental concepts in biology and medicine. Understanding these principles is essential for appreciating the intricacies of cellular function, the importance of fluid balance, and the implications for various health conditions. From intravenous fluid administration to the treatment of dehydration, the impact of hypertonicity on red blood cells is a critical factor in maintaining human health. Further research into the precise mechanisms and interactions involved will continue to shed light on this crucial aspect of cellular biology and its implications for medical practice. The knowledge gained continues to inform better diagnostic and therapeutic approaches for a variety of conditions.
Latest Posts
Latest Posts
-
Compare And Contrast Pulmonary And Systemic Circulation
Apr 03, 2025
-
Icse Tuitions Classes In Jail Road Nashik
Apr 03, 2025
-
What Is A Free Variable In A Matrix
Apr 03, 2025
-
Compare Protein Synthesis In Prokaryotes And Eukaryotes
Apr 03, 2025
-
A Closed Figure Made Of Line Segments
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about A Red Blood Cell Placed In A Hypertonic Medium Will . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
