A Horizontal Row On The Periodic Table
Juapaving
Apr 04, 2025 · 7 min read
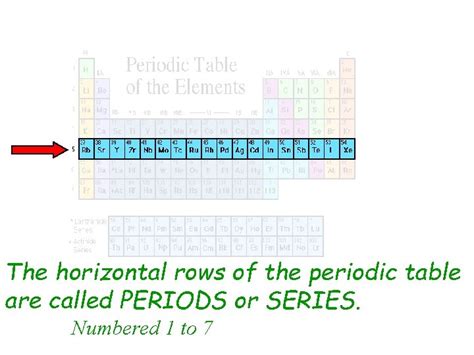
Table of Contents
A Horizontal Row on the Periodic Table: Exploring the Periods
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic number and recurring chemical properties. Understanding its structure is crucial to comprehending the behavior of matter. While columns, known as groups, share similar chemical characteristics due to identical valence electron configurations, horizontal rows, called periods, tell a different story. This article delves deep into the intricacies of a period on the periodic table, exploring the trends and patterns that emerge across these horizontal lines. We’ll examine how properties change as we move across a period, the underlying reasons for these changes, and the implications for the elements' behavior.
Understanding the Structure of a Period
Each period represents a principal energy level or shell that an atom's electrons can occupy. The first period, with just hydrogen (H) and helium (He), is the shortest, containing only elements whose electrons fill the 1s orbital. As we move down the periodic table to subsequent periods, the number of elements increases because more subshells (s, p, d, f) become available to accommodate electrons. Therefore, the length of a period reflects the number of electrons that can occupy the orbitals of a given principal energy level.
- Period 1: Contains 2 elements (Hydrogen and Helium) filling the 1s orbital.
- Period 2: Contains 8 elements (Lithium to Neon) filling the 2s and 2p orbitals.
- Period 3: Contains 8 elements (Sodium to Argon) filling the 3s and 3p orbitals.
- Period 4: Contains 18 elements (Potassium to Krypton) filling the 4s, 3d, and 4p orbitals. The inclusion of the d-block orbitals significantly increases the period's length.
- Period 5: Contains 18 elements (Rubidium to Xenon) filling the 5s, 4d, and 5p orbitals.
- Period 6: Contains 32 elements (Cesium to Radon) filling the 6s, 4f, 5d, and 6p orbitals. The addition of the f-block orbitals further expands the period.
- Period 7: Contains 32 elements (Francium to Oganesson), filling the 7s, 5f, 6d, and 7p orbitals (partially filled for some elements).
The increasing length of periods reflects the complex filling of electron orbitals, showcasing the intricate quantum mechanical nature of electron arrangement within atoms.
Periodic Trends Across a Period: A Detailed Analysis
Moving from left to right across a period, several crucial properties exhibit predictable trends. These trends are directly related to the increasing nuclear charge and the addition of electrons to the same principal energy level.
1. Atomic Radius: A Shrinking Trend
Atomic radius, the distance from the nucleus to the outermost electron, generally decreases across a period. As we move from left to right, the number of protons in the nucleus increases, resulting in a stronger positive charge. This stronger positive charge attracts the electrons more tightly, pulling them closer to the nucleus and consequently reducing the atomic radius. This effect outweighs the addition of electrons to the same energy level.
2. Ionization Energy: An Increasing Ascent
Ionization energy, the energy required to remove an electron from a neutral atom, generally increases across a period. The increased nuclear charge across a period makes it increasingly difficult to remove an electron. The stronger attraction between the nucleus and the electrons necessitates more energy to overcome this attraction and remove an electron. Therefore, ionization energy reflects the increasing hold the nucleus has on its electrons.
3. Electronegativity: A Growing Attraction
Electronegativity, the ability of an atom to attract electrons in a chemical bond, generally increases across a period. Similar to ionization energy, the increasing nuclear charge pulls the bonding electrons closer, enhancing the atom's ability to attract electrons from another atom. Elements with high electronegativity tend to form strong bonds with more electropositive elements.
4. Electron Affinity: The Energy of Electron Gain
Electron affinity, the energy change when an atom gains an electron, generally increases (becomes more negative) across a period. While not as straightforward as other trends, a more negative electron affinity generally reflects a stronger attraction for an additional electron. This is particularly evident for non-metal elements as they tend to gain electrons more readily to achieve a stable electron configuration. However, exceptions do exist, and factors beyond simple nuclear charge influence the precise value of electron affinity.
5. Metallic Character: A Gradual Decline
Metallic character, the tendency of an element to exhibit metallic properties like conductivity and malleability, generally decreases across a period. As we move from left to right, elements transition from metals to metalloids and finally nonmetals. This trend is tied to the decreasing tendency to lose electrons and the increasing ability to gain electrons. Metals readily lose electrons to achieve stable electron configurations, while nonmetals prefer to gain electrons.
6. Chemical Reactivity: A Complex Relationship
Chemical reactivity is less straightforward and depends on the element's position and electron configuration. For instance, alkali metals (Group 1) exhibit high reactivity due to their tendency to lose one electron. Halogens (Group 17) also display high reactivity due to their strong tendency to gain one electron to achieve a stable octet. Noble gases (Group 18), with full valence electron shells, are exceptionally unreactive. Therefore, reactivity is influenced by both the element's group and period.
Exceptions and Irregularities: Understanding the Nuances
While the trends described above generally hold true, several exceptions and irregularities exist. These are largely due to the subtle interplay of electron-electron repulsions, shielding effects, and the stability of half-filled and fully-filled orbitals. For example, some irregularities are observed in the ionization energies of certain elements due to the extra stability provided by half-filled or fully-filled subshells.
- Half-filled and Fully-filled Subshells: Elements with half-filled or fully-filled subshells exhibit enhanced stability, leading to slightly higher ionization energies than expected.
- Electron-Electron Repulsions: Repulsive forces between electrons can slightly counteract the attractive force of the nucleus, leading to deviations from the general trends.
- Shielding Effect: The inner electrons shield the outer electrons from the full effect of the nuclear charge, slightly reducing the effective nuclear charge experienced by outer electrons.
Understanding these subtle nuances is critical for a complete understanding of the periodic trends.
The Significance of Periods in Chemical Bonding and Reactions
The period an element belongs to directly impacts its chemical behavior and the types of bonds it forms. Elements within the same period have similar principal quantum numbers but varying numbers of valence electrons. This difference leads to diverse chemical reactivities.
- Bonding: Elements in the same period can form various types of bonds, ranging from ionic bonds (formed between metals and nonmetals) to covalent bonds (formed between nonmetals). The electronegativity difference between elements in a given period influences the type of bond formed.
- Reactions: The reactivity of elements within a period varies significantly. Metals generally react by losing electrons, while nonmetals gain electrons. The reactivity is influenced by their position relative to stable electron configurations (noble gases).
- Oxidation States: The possible oxidation states of elements within a period are influenced by the number of valence electrons they possess and their ability to gain or lose electrons.
Applications and Relevance
Understanding the trends within a period has far-reaching applications in various fields:
- Material Science: The properties of elements within a period dictate the characteristics of materials made from them. For example, the transition metals in period 4 exhibit a wide range of magnetic and catalytic properties.
- Chemical Synthesis: Knowledge of periodic trends allows chemists to predict the behavior of elements during chemical reactions and helps design synthetic pathways for creating new compounds.
- Drug Discovery: The properties of elements within a period play a crucial role in determining the activity and toxicity of drugs. Understanding these properties is essential in the design and development of new pharmaceuticals.
- Environmental Science: The reactivity of elements within a period determines their environmental behavior and their impact on ecosystems.
Conclusion: A Comprehensive Perspective
The horizontal rows on the periodic table, the periods, offer a wealth of information about the properties and behaviors of elements. The predictable trends in atomic radius, ionization energy, electronegativity, electron affinity, and metallic character are crucial for understanding chemical behavior. However, it's equally important to recognize the exceptions and irregularities that arise due to the complexities of electron configurations and interactions within atoms. The knowledge gained from studying the periodic trends within a period underpins our understanding of chemical bonding, reactivity, and the diverse applications of elements in various fields. A deep understanding of periods provides a fundamental framework for advancing scientific research and technological innovations across multiple disciplines.
Latest Posts
Latest Posts
-
The Speed Of Light In A Vacuum Is
Apr 04, 2025
-
What Is The Largest Animal On Land
Apr 04, 2025
-
Find The Prime Factorization Of 75
Apr 04, 2025
-
A Group Of Cells That Perform Similar Functions
Apr 04, 2025
-
How Is Weathering Different From Erosion
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about A Horizontal Row On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
