Which Of The Following Is Stronger Acid
Juapaving
Apr 05, 2025 · 6 min read
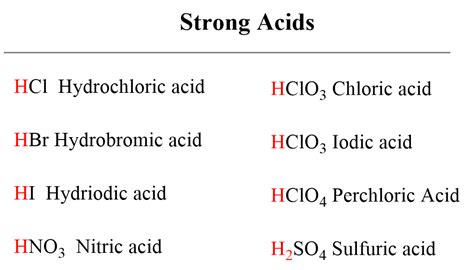
Table of Contents
Which is the Stronger Acid? A Deep Dive into Acid Strength
Determining which of two acids is stronger involves understanding the factors that influence acidity. This isn't simply about memorizing a list; it's about grasping the underlying chemical principles. This comprehensive guide will explore various aspects of acid strength, providing you with the tools to confidently compare acids and predict their relative strengths.
What Makes an Acid Strong?
The strength of an acid is determined by its ability to donate a proton (H⁺) to a base. A strong acid readily donates its proton, resulting in complete or near-complete dissociation in water. Conversely, a weak acid only partially dissociates, meaning a significant portion of the acid remains in its undissociated form in solution.
This dissociation can be represented by the following general equation:
HA ⇌ H⁺ + A⁻
Where:
- HA represents the acid
- H⁺ represents the proton (hydrogen ion)
- A⁻ represents the conjugate base
The equilibrium constant for this reaction, denoted as Ka (acid dissociation constant), is a quantitative measure of acid strength. A larger Ka value indicates a stronger acid, as it signifies a greater tendency to donate protons. Conversely, a smaller Ka value indicates a weaker acid. Often, pKa (the negative logarithm of Ka) is used; a smaller pKa value indicates a stronger acid.
Factors Affecting Acid Strength:
Several factors influence the strength of an acid:
1. Bond Strength:
The strength of the bond between the hydrogen atom and the rest of the acid molecule plays a crucial role. A weaker H-A bond leads to easier proton donation and thus a stronger acid. This is because less energy is required to break the bond.
Consider HCl (hydrochloric acid) and HF (hydrofluoric acid). The H-Cl bond is significantly weaker than the H-F bond. As a result, HCl is a much stronger acid than HF. The strong electronegativity of fluorine pulls the electron density towards itself, strengthening the H-F bond and making proton donation more difficult.
2. Electronegativity:
The electronegativity of the atom bonded to the hydrogen significantly impacts acid strength. A more electronegative atom attracts the shared electrons in the H-A bond more strongly, weakening the bond and facilitating proton donation. This explains why oxyacids (acids containing oxygen) tend to be stronger than their non-oxy counterparts.
For instance, compare HClO₄ (perchloric acid) and HClO (hypochlorous acid). Perchloric acid has more oxygen atoms bonded to the chlorine atom, increasing the overall electronegativity and making the O-H bond weaker. Consequently, HClO₄ is a significantly stronger acid than HClO.
3. Size of the Anion:
The size of the conjugate base (A⁻) formed after proton donation influences acid strength. A larger, more stable conjugate base is more easily formed, leading to a stronger acid. This is because a larger anion can better distribute the negative charge, reducing its overall energy.
For example, consider HBr and HI. Both are strong acids, but HI is slightly stronger. The iodide ion (I⁻) is larger than the bromide ion (Br⁻), making it better able to stabilize the negative charge, thus facilitating proton donation.
4. Resonance:
The presence of resonance structures in the conjugate base can significantly enhance acid strength. Resonance allows for delocalization of the negative charge over multiple atoms, stabilizing the conjugate base and making proton donation more favorable.
Carboxylic acids (RCOOH) are a prime example. The carboxylate anion (RCOO⁻) formed upon proton donation exhibits resonance stabilization, making carboxylic acids relatively strong acids compared to alcohols (ROH).
5. Inductive Effect:
Electron-withdrawing groups (groups that pull electron density away from the acidic hydrogen) increase acid strength. This effect is particularly relevant in organic acids. The more electron-withdrawing groups present, the weaker the O-H bond becomes, and the stronger the acid becomes.
Consider the effect of chlorine atoms on acetic acid (CH₃COOH). As the number of chlorine atoms increases (e.g., monochloroacetic acid, dichloroacetic acid, trichloroacetic acid), the acid strength dramatically increases due to the strong electron-withdrawing effect of chlorine.
Comparing Specific Acids:
Let's analyze a few common scenarios to illustrate how these factors determine acid strength.
Scenario 1: HCl vs. HF
As discussed earlier, HCl is a much stronger acid than HF. This is primarily due to the weaker H-Cl bond compared to the H-F bond. The higher electronegativity of fluorine actually strengthens the H-F bond, making proton donation less favorable.
Scenario 2: H₂SO₄ vs. H₂SO₃
Sulfuric acid (H₂SO₄) is a much stronger acid than sulfurous acid (H₂SO₃). This difference arises from the additional oxygen atom in sulfuric acid. The extra oxygen atom increases the electronegativity around the sulfur atom, weakening the O-H bonds and making proton donation easier. This is a clear example of the impact of electronegativity and the number of oxygen atoms in oxyacids.
Scenario 3: CH₃COOH vs. CH₃CH₂OH
Acetic acid (CH₃COOH) is a stronger acid than ethanol (CH₃CH₂OH). This is because the acetate anion (CH₃COO⁻) formed upon proton donation exhibits resonance stabilization, significantly increasing its stability. The ethoxide anion (CH₃CH₂O⁻), on the other hand, lacks this resonance stabilization.
Scenario 4: HNO₃ vs. HNO₂
Nitric acid (HNO₃) is a stronger acid than nitrous acid (HNO₂). The additional oxygen atom in nitric acid increases its electronegativity, weakening the O-H bond and promoting proton donation. The resonance structures also contribute to the increased stability of the nitrate anion (NO₃⁻) compared to the nitrite anion (NO₂⁻).
Predicting Acid Strength: A Practical Approach
While a detailed quantitative analysis using Ka or pKa values provides the most precise comparison, a qualitative approach can often suffice for predicting relative acid strength. Consider the following steps:
- Identify the acidic hydrogen: Pinpoint the hydrogen atom that will be donated as a proton.
- Assess bond strength: Evaluate the strength of the bond between the acidic hydrogen and the rest of the molecule. Weaker bonds generally indicate stronger acids.
- Analyze electronegativity: Consider the electronegativity of the atom bonded to the acidic hydrogen. Higher electronegativity leads to weaker bonds and stronger acids.
- Examine resonance: Assess whether resonance stabilization exists in the conjugate base. Greater resonance stabilization leads to a stronger acid.
- Consider inductive effects: Identify electron-withdrawing or electron-donating groups attached to the molecule. Electron-withdrawing groups enhance acid strength.
- Evaluate conjugate base size: A larger, more stable conjugate base indicates a stronger acid.
By systematically analyzing these factors, you can effectively compare the strengths of different acids, even without knowing their precise Ka or pKa values.
Conclusion:
Determining which acid is stronger involves a multifaceted evaluation of various chemical principles. While memorizing a list of acids and their strengths is helpful, understanding the underlying factors—bond strength, electronegativity, resonance, inductive effects, and conjugate base stability—empowers you to make informed predictions and deepen your understanding of acid-base chemistry. This approach not only helps you solve specific problems but also fosters a more robust comprehension of the subject. Remember, the strength of an acid is not just a property; it's a reflection of its inherent chemical nature and the interplay of several crucial factors.
Latest Posts
Latest Posts
-
Equation Of A Plane Through 3 Points
Apr 05, 2025
-
An Ex Policeman Lost His House Riddle
Apr 05, 2025
-
Focal Length Of Convex Lens Is Positive Or Negative
Apr 05, 2025
-
One Six Letter Word To Describe Your Daughter In English
Apr 05, 2025
-
Name Of A Seven Sided Shape
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Is Stronger Acid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
