Which Of The Following Is A Strongest Acid
Juapaving
Apr 03, 2025 · 5 min read
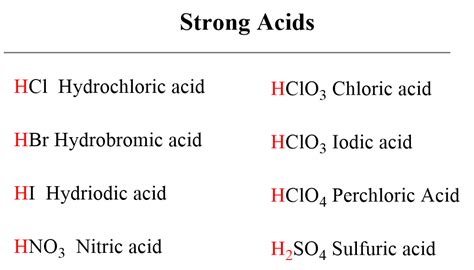
Table of Contents
Which of the Following is the Strongest Acid? Understanding Acid Strength
Determining the strongest acid from a given set requires a deep understanding of acid-base chemistry. It's not simply a matter of looking at the chemical formula; several factors influence an acid's strength. This article delves into the intricacies of acid strength, exploring the key concepts and providing a framework for comparing and contrasting different acids. We will cover the definitions of acids and bases, delve into the factors affecting acid strength, and finally, provide a methodology for determining the strongest acid among a given group.
Defining Acids and Bases: A Necessary Foundation
Before we can discuss which acid is strongest, we must establish a clear understanding of what constitutes an acid. While several definitions exist (Arrhenius, Brønsted-Lowry, Lewis), the Brønsted-Lowry definition proves most useful for comparing acid strength:
- Brønsted-Lowry Acid: A substance that donates a proton (H⁺).
The strength of an acid is directly related to its ability to donate this proton. A strong acid readily donates its proton in aqueous solution, while a weak acid only partially dissociates, meaning it holds onto its proton more tightly. The strength of an acid is quantified using its acid dissociation constant (Ka).
- Ka: A quantitative measure of acid strength. A higher Ka value indicates a stronger acid because it signifies a greater extent of proton donation. Often, the negative logarithm of Ka (pKa = -log Ka) is used; a lower pKa indicates a stronger acid.
Factors Influencing Acid Strength: A Deeper Dive
Several factors contribute to an acid's ability to donate a proton:
1. Electronegativity: The electronegativity of the atom bonded to the acidic hydrogen plays a crucial role. A more electronegative atom pulls electron density away from the O-H bond, weakening it and making proton donation easier. This is why, for example, HCl is a stronger acid than H₂S; chlorine is significantly more electronegative than sulfur.
2. Bond Strength: The strength of the bond between the hydrogen atom and the rest of the molecule impacts acid strength. A weaker bond facilitates easier proton release, leading to a stronger acid. In general, shorter bonds are stronger, and longer bonds are weaker.
3. Resonance Stabilization: The stability of the conjugate base (the species remaining after the acid donates its proton) heavily influences acid strength. If the conjugate base is stabilized through resonance (delocalization of electron density), the acid will be stronger. This is because the negative charge is spread across multiple atoms, making the conjugate base more stable. Carboxylic acids, for example, exhibit resonance stabilization, making them relatively strong acids compared to alcohols.
4. Inductive Effects: Electron-withdrawing groups (EWGs) attached to the molecule can increase acid strength through an inductive effect. These groups pull electron density away from the O-H bond, making proton donation easier. Conversely, electron-donating groups (EDGs) weaken the acid by increasing electron density around the O-H bond.
5. Solvent Effects: The solvent in which the acid is dissolved can significantly influence its apparent strength. Protic solvents (those containing O-H or N-H bonds) can stabilize the conjugate base, thus increasing the acid's strength. The strength of an acid can change depending on the solvent used.
6. Size and Polarizability: Larger atoms are more polarizable, meaning their electron clouds are more easily distorted. This increased polarizability can weaken the bond to hydrogen, resulting in a stronger acid. This effect is particularly noticeable when comparing acids within the same group of the periodic table. For example, HI is a stronger acid than HF because iodine is larger and more polarizable than fluorine.
Comparing Acids: A Step-by-Step Approach
To determine the strongest acid from a given set, follow these steps:
- Identify the acidic proton: Locate the hydrogen atom(s) that can be donated.
- Consider electronegativity: Compare the electronegativities of the atoms bonded to the acidic hydrogen. Higher electronegativity generally leads to stronger acidity.
- Assess bond strength: Analyze the bond strength between the hydrogen and the rest of the molecule. Weaker bonds indicate stronger acids.
- Examine resonance stabilization: Determine if the conjugate base can be stabilized by resonance. More extensive resonance leads to stronger acidity.
- Evaluate inductive effects: Consider the presence of electron-withdrawing or electron-donating groups. EWGs increase acidity, while EDGs decrease it.
- Account for solvent effects (if applicable): If the acids are dissolved in different solvents, their relative strengths may vary.
- Utilize pKa values (if available): If pKa values are provided, the acid with the lowest pKa is the strongest.
Example: Comparing Acid Strengths
Let's compare the following acids: HCl, HF, HBr, and HI.
- Acidic proton: All four acids have a single acidic proton.
- Electronegativity: Fluorine is the most electronegative, followed by chlorine, bromine, and iodine.
- Bond strength: The bond strength decreases down the group (H-F > H-Cl > H-Br > H-I).
- Resonance: No resonance stabilization is involved in these acids.
- Inductive effects: No significant inductive effects are present.
- Solvent effects: Assuming all are in the same solvent (e.g., water), this factor is consistent.
Based on bond strength, the order of acid strength would be HF < HCl < HBr < HI. While fluorine is the most electronegative, the exceptionally strong H-F bond outweighs this effect, making HI the strongest acid among this group.
Conclusion: Understanding the Nuances of Acid Strength
Determining the strongest acid requires a comprehensive consideration of multiple factors. Simply relying on a single parameter, such as electronegativity, can be misleading. A holistic approach, encompassing electronegativity, bond strength, resonance, inductive effects, and solvent effects, provides a robust framework for comparing and contrasting the strengths of different acids. By systematically evaluating these factors, one can confidently identify the strongest acid among a given set. Remember to consult pKa values when available, as they provide a direct quantitative measure of acid strength. This detailed understanding of acid strength is crucial for various applications in chemistry and related fields.
Latest Posts
Latest Posts
-
Every Rectangle Is A Square True Or False
Apr 04, 2025
-
Five Letter Words With Two A
Apr 04, 2025
-
Label The Parts Of The Animal Cell
Apr 04, 2025
-
Land Surrounded By Water On 3 Sides
Apr 04, 2025
-
Compare And Contrast Mixtures And Solutions
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Is A Strongest Acid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
