What Is The State Of Matter Of Fire
Juapaving
Apr 02, 2025 · 5 min read
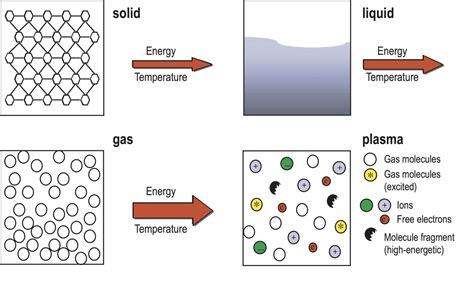
Table of Contents
What is the State of Matter of Fire?
The question, "What is the state of matter of fire?" seems simple enough, but it reveals a surprisingly complex answer. While we intuitively understand fire as something we can see and interact with, classifying it neatly into one of the traditional states of matter – solid, liquid, gas, or plasma – proves challenging. The truth is, fire isn't a state of matter itself; it's a process, a chemical reaction. Understanding fire requires delving into the intricacies of combustion and the various states of matter involved.
The Chemistry of Combustion: A Complex Process
Fire, at its core, is a rapid oxidation process – a chemical reaction between a fuel source and an oxidizer, usually oxygen. This reaction releases energy in the form of heat and light. The intensity and characteristics of the fire depend on several factors, including:
-
The fuel source: This can be anything from wood and paper to gases like methane and propane. Different fuels have different combustion properties, influencing the temperature and appearance of the fire.
-
The oxidizer: Oxygen is the most common oxidizer, but other substances can support combustion.
-
The temperature: A certain activation energy is required to initiate combustion. Once started, the heat released sustains the reaction.
-
The chain reaction: Combustion often involves a chain reaction, where the heat generated produces more reactive species, propagating the fire.
The Four Stages of Combustion
The combustion process itself can be broadly divided into four stages:
-
Preheating: The fuel is heated to its ignition temperature.
-
Ignition: The fuel reaches a point where it spontaneously ignites and begins to burn.
-
Flaming combustion: This is the sustained burning phase characterized by the visible flames.
-
Glowing combustion: After the flammable gases are consumed, the remaining fuel smolders, emitting heat and light.
States of Matter Involved in Fire
While fire itself isn't a state of matter, various states of matter are involved in the combustion process. Let's break down the roles of each:
1. Solid Phase: Fuel Source
The initial fuel often exists as a solid. Think of a burning log or a piece of paper. The heat from the fire causes the solid fuel to decompose, releasing volatile gases that then participate in the combustion reaction. The solid residue left behind, such as ash, represents the remaining non-combustible portion.
2. Liquid Phase: Fuel & Products
Some fuels are liquids, like gasoline or alcohol. Liquids evaporate to form gaseous fuels which readily participate in combustion. Furthermore, certain combustion products might condense into a liquid state during the cooling phase after the fire is extinguished.
3. Gas Phase: The Heart of Combustion
The gaseous phase is crucial for fire. This is where the primary combustion reaction occurs. Gaseous fuel molecules combine with oxygen in the air, releasing energy as heat and light. The flames themselves are primarily composed of hot, glowing gases and reactive species. The specific composition of these gases depends largely on the nature of the fuel.
4. Plasma Phase: A High-Energy State
At sufficiently high temperatures, the gases involved in combustion can ionize, transitioning into a plasma state. Plasma is a highly energized state of matter where electrons are stripped from atoms, resulting in a mixture of positively charged ions and free electrons. While not always dominant in everyday fires, the presence of plasma significantly contributes to the intensity and characteristics of the flames, especially in high-temperature combustion processes like those found in welding torches or stars.
Why Fire Isn't a State of Matter
Fire's dynamic nature makes it distinct from the traditional states of matter. It is not a substance with a defined chemical composition and fixed structure. Instead, it's a phenomenon, a process governed by chemical reactions. To further illustrate this point:
-
No fixed chemical composition: Fire's composition varies depending on the fuel, oxidizer, and the conditions of combustion. There's no single chemical formula representing "fire."
-
Dynamic nature: Fire is constantly changing. The temperature, the composition of gases, and the rate of reaction are all in flux. A static state of matter maintains a more or less consistent structure and composition.
-
Process, not a substance: Fire is the visible manifestation of a rapid exothermic reaction. It's the energy release and transformation that we perceive, not a static substance.
The Role of Light and Heat in Fire
The heat and light emitted by fire are crucial components of the phenomenon. The intense heat drives the chemical reaction, perpetuating the combustion process. The light emitted comes from the excited atoms and molecules within the flames. Different elements in the fuel source produce characteristic colors, aiding in fire identification and analysis.
Temperature and Flame Color
The temperature of a flame significantly impacts its color. Cooler flames often appear orange or red, while hotter flames can be blue or white. This color variation is due to the different wavelengths of light emitted at various temperatures by the excited particles in the flame.
Misconceptions about Fire
Several common misconceptions surround the nature of fire:
-
Fire is not a living thing: It lacks the characteristics of life, such as reproduction, metabolism, and growth. It's a purely chemical and physical process.
-
Fire doesn't have a definite shape: The shape of a flame is determined by the surrounding environment, including airflow and the nature of the fuel source. It's not an inherent characteristic of fire itself.
-
Fire is not a single element: Fire isn't composed of a single chemical element; it's a complex mixture of reacting gases, ions, and energized particles.
Conclusion: A Process, Not a State
In summary, fire is not a state of matter; it's a process, specifically a chemical reaction involving the rapid oxidation of a fuel source. Although it involves multiple states of matter—solid, liquid, gas, and potentially plasma—the phenomenon of fire itself is a dynamic interplay of chemical and physical changes. Understanding the chemistry of combustion, the role of different states of matter, and the energy transformations involved gives a comprehensive understanding of this fascinating and powerful natural phenomenon. The continuous research and study of fire remains crucial to understanding its complexities, from the development of safer fire prevention techniques to improving the efficiency of energy production processes. Continued research into the multifaceted nature of fire promises to reveal even more intricate details about this seemingly simple yet captivating phenomenon.
Latest Posts
Latest Posts
-
Balaneced Chemical Reaction Between C4h10 Oz Yeliding Co2
Apr 03, 2025
-
Which Is The Longest Phase In Meiosis
Apr 03, 2025
-
How Many Commas In A Billion
Apr 03, 2025
-
Glycolysis Ends In The Production Of
Apr 03, 2025
-
Distributive Law 3 Variables Discrete Math
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The State Of Matter Of Fire . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
