What Is The Electron Configuration Of Bromine
Juapaving
Apr 05, 2025 · 6 min read
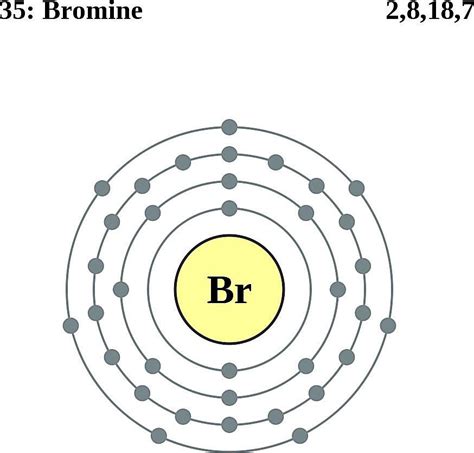
Table of Contents
What is the Electron Configuration of Bromine? A Deep Dive into Atomic Structure
Bromine, a vibrant reddish-brown liquid element, holds a fascinating place in the periodic table. Understanding its electron configuration is key to unlocking its unique chemical properties and reactivity. This comprehensive guide will delve into the intricacies of bromine's electron configuration, exploring its implications for its behavior and applications.
Understanding Electron Configuration
Before we dive into bromine specifically, let's establish a foundational understanding of electron configuration. An element's electron configuration describes how electrons are distributed among the various energy levels (shells) and sublevels (subshells) within an atom. This arrangement dictates how an atom will interact with other atoms, forming chemical bonds and determining its chemical properties.
The electron configuration follows specific rules:
- Aufbau Principle: Electrons fill orbitals starting with the lowest energy levels and progressing upwards.
- Pauli Exclusion Principle: Each orbital can hold a maximum of two electrons, each with opposite spins.
- Hund's Rule: Electrons individually occupy each orbital within a subshell before pairing up.
These rules help us predict the most stable electron configuration for an atom. We represent electron configurations using a shorthand notation, specifying the principal quantum number (n), the type of subshell (s, p, d, or f), and the number of electrons in each subshell.
Determining Bromine's Atomic Number and Electron Count
Bromine (Br) is located in Group 17 (halogens) and Period 4 of the periodic table. Its atomic number is 35, which means a neutral bromine atom contains 35 protons and 35 electrons. This electron count is crucial for determining its electron configuration.
The Electron Configuration of Bromine
Using the Aufbau principle, Hund's rule, and the Pauli exclusion principle, we can systematically fill the orbitals of a bromine atom:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁵
Let's break down this configuration:
- 1s²: The first energy level (n=1) contains one subshell, the s subshell, which holds two electrons.
- 2s² 2p⁶: The second energy level (n=2) contains two subshells: the s subshell (two electrons) and the p subshell (six electrons).
- 3s² 3p⁶: The third energy level (n=3) also contains s and p subshells, each filled to their maximum capacity.
- 4s²: The fourth energy level begins with the s subshell, which holds two electrons.
- 3d¹⁰: Before proceeding to the 4p subshell, the 3d subshell fills completely with ten electrons. This illustrates that energy levels can overlap slightly.
- 4p⁵: The 4p subshell contains five electrons, leaving one space unfilled. This incomplete p-subshell is crucial to bromine's reactivity.
Noble Gas Configuration and Shorthand Notation
To simplify the electron configuration, we often use the noble gas configuration. Bromine follows argon (Ar) in the periodic table, so we can represent its configuration using argon's core configuration:
[Ar] 4s² 3d¹⁰ 4p⁵
This shorthand notation concisely expresses that bromine possesses the same electron configuration as argon up to the 3p subshell, and then adds electrons to the 4s, 3d, and 4p subshells.
Implications of Bromine's Electron Configuration
Bromine's electron configuration, particularly the incomplete 4p subshell, directly influences its chemical properties:
- High Electronegativity: With seven valence electrons (electrons in the outermost shell), bromine has a strong tendency to gain one electron to achieve a stable octet (eight valence electrons) – a configuration similar to the noble gas krypton. This high electronegativity makes bromine highly reactive.
- Formation of Halide Ions: Bromine readily accepts an electron to form the bromide ion (Br⁻), which has a stable octet configuration. This ion is found in various ionic compounds.
- Covalent Bonding: Bromine can also form covalent bonds by sharing electrons with other atoms, particularly nonmetals. This is evident in diatomic bromine (Br₂), where two bromine atoms share a pair of electrons to achieve a stable octet.
- Oxidizing Agent: Because of its tendency to gain electrons, bromine acts as a strong oxidizing agent, meaning it can readily accept electrons from other substances.
Bromine's Valence Electrons and Chemical Reactivity
The valence electrons, those in the outermost shell (n=4 in bromine's case), play a pivotal role in chemical bonding and reactivity. Bromine's seven valence electrons (4s² 4p⁵) explain its strong tendency to gain an electron, forming a -1 ion. This tendency dictates its reactivity with metals (forming ionic compounds) and nonmetals (forming covalent compounds).
Applications of Bromine and its Compounds
Bromine's unique properties, dictated by its electron configuration, lead to a wide range of applications:
- Flame Retardants: Brominated compounds are used as flame retardants in plastics, textiles, and other materials. Their effectiveness stems from their ability to interfere with combustion processes.
- Water Treatment: Bromine compounds, such as sodium bromate, are used in water treatment to disinfect water and control algae growth.
- Agricultural Chemicals: Some bromine-containing compounds are used as pesticides and fumigants. However, the environmental impact of these applications is a growing concern.
- Medical Applications: Certain bromine compounds have medicinal applications, such as in sedatives and anticonvulsants.
- Photography: Silver bromide is a key component in photographic film and paper, reacting to light to create images.
- Dyes and Pigments: Bromine compounds are used in the manufacture of some dyes and pigments.
Safety Concerns and Environmental Impact
While bromine and its compounds have many practical applications, it’s crucial to consider their potential safety and environmental impacts. Bromine is a corrosive and toxic substance, requiring careful handling. Certain brominated compounds, like some flame retardants, have been shown to persist in the environment and accumulate in living organisms, potentially causing health problems. Ongoing research is focused on developing safer alternatives.
Further Exploration
Understanding bromine's electron configuration provides a foundation for understanding its behavior and properties. To deepen your understanding, you can explore more advanced concepts such as:
- Molecular Orbital Theory: This theory provides a more sophisticated model of bonding in molecules, including diatomic bromine.
- Spectroscopy: Techniques like UV-Vis spectroscopy can provide experimental evidence that supports our understanding of bromine's electron configuration.
- Computational Chemistry: Modern computational methods allow us to calculate and predict the properties of bromine-containing molecules with high accuracy.
Conclusion
The electron configuration of bromine, 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁵ or [Ar] 4s² 3d¹⁰ 4p⁵, is the key to understanding its chemical behavior. Its seven valence electrons, particularly the incomplete 4p subshell, drive its reactivity as a powerful oxidizing agent, leading to its many applications in diverse fields. However, it is essential to consider the safety and environmental aspects of bromine and its compounds, promoting responsible usage and research into sustainable alternatives. By understanding the fundamental principles of atomic structure and electron configuration, we can appreciate the multifaceted nature of bromine and its role in the world around us.
Latest Posts
Latest Posts
-
Eclipses Do Not Occur Every Month Because The Moons
Apr 05, 2025
-
Do Lipids Store More Energy Than Carbohydrates
Apr 05, 2025
-
Difference Between Real Numbers And Natural Numbers
Apr 05, 2025
-
What Does Vitamin A B C And D Do
Apr 05, 2025
-
How To Write 1850 00 On A Check
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Is The Electron Configuration Of Bromine . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
