What Is The Chemical Formula Of Protein
Juapaving
Apr 02, 2025 · 6 min read
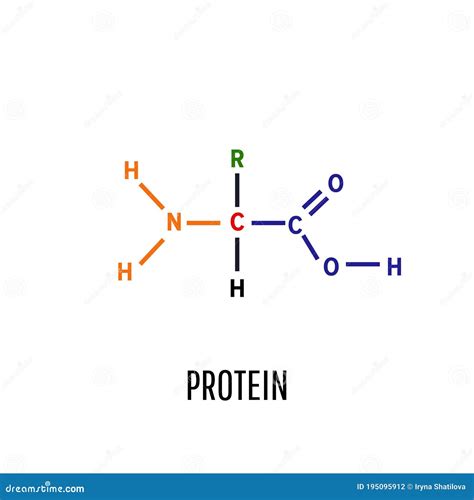
Table of Contents
What is the Chemical Formula of Protein? A Deep Dive into Amino Acid Chains
Proteins are the workhorses of life, crucial for virtually every biological process. From catalyzing reactions to providing structural support, their multifaceted roles are essential for the proper functioning of all living organisms. But what exactly is a protein, chemically speaking? Understanding the chemical formula of a protein isn't as straightforward as, say, the formula for water (H₂O). This is because proteins aren't composed of a single, uniform molecule, but rather a vast array of different molecules, each with its unique sequence and structure.
The Building Blocks: Amino Acids
The fundamental building blocks of all proteins are amino acids. These organic molecules contain a central carbon atom (the alpha carbon) bonded to four groups:
- An amino group (-NH₂): This is a basic group, meaning it can accept a proton (H⁺).
- A carboxyl group (-COOH): This is an acidic group, meaning it can donate a proton (H⁺).
- A hydrogen atom (-H): A simple hydrogen atom.
- A side chain (R-group): This is what distinguishes one amino acid from another. The R-group can be anything from a simple hydrogen atom (as in glycine) to a complex aromatic ring structure (as in tryptophan).
There are 20 standard amino acids that are encoded by the genetic code and used to build proteins in living organisms. Each of these amino acids has a specific chemical formula based on its unique R-group.
Examples of Amino Acid Chemical Formulas:
- Glycine (Gly, G): NH₂CH₂COOH
- Alanine (Ala, A): CH₃CH(NH₂)COOH
- Serine (Ser, S): HOCH₂CH(NH₂)COOH
- Aspartic Acid (Asp, D): HOOCCH₂CH(NH₂)COOH
- Phenylalanine (Phe, F): C₆H₅CH₂CH(NH₂)COOH
These are just a few examples, and each of the 20 standard amino acids has its own unique chemical formula. The complexity arises from the variability of the R-group. Some R-groups are nonpolar and hydrophobic (water-fearing), others are polar and hydrophilic (water-loving), and some carry a net positive or negative charge at physiological pH. This diversity in R-groups is crucial for the vast array of functions that proteins can perform.
The Peptide Bond: Linking Amino Acids
Amino acids are linked together through a peptide bond, a type of covalent bond formed between the carboxyl group of one amino acid and the amino group of another. This reaction involves the removal of a water molecule (dehydration synthesis). The resulting chain of amino acids is called a polypeptide. A protein is essentially one or more polypeptides folded into a specific three-dimensional structure.
The Peptide Bond Formation Reaction:
The general reaction can be represented as:
R₁-COOH + H₂N-R₂ → R₁-CO-NH-R₂ + H₂O
Where:
- R₁ represents the side chain of the first amino acid.
- R₂ represents the side chain of the second amino acid.
The peptide bond (-CO-NH-) is a strong covalent bond, essential for maintaining the integrity of the polypeptide chain. The sequence of amino acids in a polypeptide chain is known as its primary structure, which dictates the subsequent higher-order structures.
Protein Structure: Beyond the Simple Formula
The chemical formula of a protein cannot simply be represented as a sum of the individual amino acid formulas because the protein's function is intimately tied to its three-dimensional structure. The higher-order structures are critical:
-
Secondary Structure: This refers to local folding patterns within the polypeptide chain, such as alpha-helices and beta-sheets. These structures are stabilized by hydrogen bonds between the amino acid backbone atoms.
-
Tertiary Structure: This is the overall three-dimensional arrangement of a single polypeptide chain. This structure is maintained by various interactions between the R-groups of the amino acids, including:
- Disulfide bonds: Covalent bonds between cysteine residues.
- Hydrogen bonds: Weaker bonds between polar R-groups.
- Ionic bonds (salt bridges): Electrostatic interactions between charged R-groups.
- Hydrophobic interactions: Clustering of nonpolar R-groups in the protein core.
-
Quaternary Structure: This refers to the arrangement of multiple polypeptide chains (subunits) to form a functional protein complex. Examples include hemoglobin and many enzymes.
Determining the Chemical Formula: A Challenging Task
Determining the precise chemical formula of a protein is a formidable task due to its complexity. While we can determine the amino acid sequence using techniques like mass spectrometry and Edman degradation, calculating a comprehensive formula considering all atoms (C, H, O, N, S, and potentially others depending on the R-groups) in a folded protein is extraordinarily complex.
The size and complexity of proteins vary enormously. Small proteins might contain only a few dozen amino acids, while others can consist of thousands. This wide range makes it impractical to represent a generalized chemical formula for all proteins.
The Importance of Protein Structure in Function
The three-dimensional structure of a protein is paramount to its function. A slight change in the amino acid sequence (a mutation) can lead to a significant alteration in the protein's structure and, consequently, its function. This can have profound effects on the organism, as seen in various genetic diseases.
For example, sickle cell anemia is caused by a single amino acid substitution in the beta-globin subunit of hemoglobin. This seemingly minor change leads to a dramatic alteration in the shape of the hemoglobin molecule, causing red blood cells to become sickle-shaped and impairing their ability to carry oxygen effectively.
Applications and Future Directions
Understanding protein structure and function is crucial in various fields, including:
- Drug discovery: Identifying and designing drugs that interact with specific proteins to treat diseases.
- Biotechnology: Engineering proteins with novel properties for industrial applications.
- Diagnostics: Developing diagnostic tools to detect and monitor diseases based on protein levels or modifications.
- Food science: Improving the nutritional value and processing of food products.
Advancements in techniques like X-ray crystallography, NMR spectroscopy, and cryo-electron microscopy are constantly improving our ability to determine the structures of increasingly complex proteins. This is vital to further unravel the intricate relationships between protein structure, function, and disease.
Conclusion
While a simple, generalized chemical formula for all proteins is impossible due to their incredible diversity, understanding the building blocks (amino acids) and the nature of peptide bonds is fundamental to understanding protein structure and function. The intricate folding patterns and interactions among amino acid side chains ultimately determine a protein's unique biological role. Continued research in protein chemistry and structural biology will undoubtedly lead to further breakthroughs in diverse fields, benefiting both human health and technological advancements. The quest to decipher the complexities of proteins continues to be a fascinating and vital area of scientific inquiry.
Latest Posts
Latest Posts
-
Biotic Factors In An Aquatic Ecosystem
Apr 03, 2025
-
Least Common Multiple Of 10 And 8
Apr 03, 2025
-
Four Ways To Write A Number
Apr 03, 2025
-
How To Calculate The Perimeter Of A Rhombus
Apr 03, 2025
-
Is The Electromagnetic Spectrum Transverse Or Longitudinal
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Chemical Formula Of Protein . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
