Vertical Columns Of The Periodic Table Are Called
Juapaving
Apr 03, 2025 · 6 min read
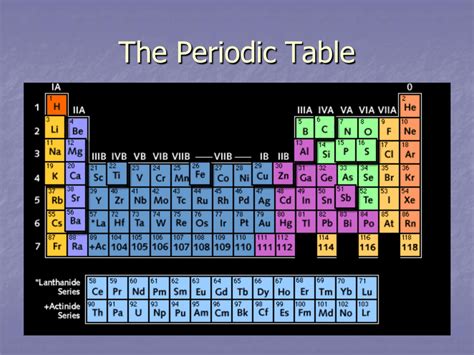
Table of Contents
Vertical Columns of the Periodic Table are Called Groups or Families: A Deep Dive
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Understanding its organization is crucial for grasping chemical behavior and predicting reactions. A common question, especially for beginners, centers around the vertical columns: what are the vertical columns of the periodic table called? The answer is simple yet profound: they are called groups or families. These columns represent elements with similar outer electron shell configurations, leading to shared chemical properties. This article will delve into the intricacies of these groups, exploring their characteristics, trends, and significance in the broader context of chemistry.
Understanding the Periodic Table's Structure
Before diving into the specifics of groups, let's establish a fundamental understanding of the periodic table's structure. The table arranges elements in a grid, with elements organized by increasing atomic number (the number of protons in an atom's nucleus). The arrangement isn't arbitrary; it reflects the periodic recurrence of similar chemical properties. The table features two main organizational components:
-
Periods (Rows): These horizontal rows represent elements with the same number of electron shells. As you move across a period, the number of electrons in the outermost shell increases, leading to gradual changes in properties.
-
Groups (Families) (Columns): These vertical columns represent elements with the same number of valence electrons (electrons in the outermost shell). Elements within the same group exhibit similar chemical behaviors due to this shared electronic configuration.
The Significance of Valence Electrons
The reason elements within the same group share similar properties boils down to valence electrons. These outermost electrons are the primary players in chemical bonding, determining how an atom interacts with other atoms. Elements in the same group have the same number of valence electrons, leading to similar bonding patterns and resulting chemical characteristics. This is why elements within a group often exhibit similar reactivity, forming similar types of compounds.
Exploring the Major Groups of the Periodic Table
The periodic table comprises 18 groups, each with unique characteristics. Let's explore some of the most important groups:
Group 1: Alkali Metals
The alkali metals (lithium, sodium, potassium, rubidium, cesium, and francium) are highly reactive metals with one valence electron. Their reactivity stems from their tendency to lose this electron to achieve a stable electron configuration. They react vigorously with water, producing hydrogen gas and forming alkaline solutions. This reactivity increases as you move down the group.
Group 2: Alkaline Earth Metals
Alkaline earth metals (beryllium, magnesium, calcium, strontium, barium, and radium) possess two valence electrons. They are also reactive metals, but less so than alkali metals. They react with water, but generally less vigorously than alkali metals. They're essential for various biological processes, with calcium and magnesium playing vital roles in living organisms.
Group 17: Halogens
Halogens (fluorine, chlorine, bromine, iodine, and astatine) are highly reactive nonmetals with seven valence electrons. They readily gain one electron to achieve a stable octet, making them strong oxidizing agents. Their reactivity decreases as you move down the group. Halogens form many important compounds, including salts and various organic molecules.
Group 18: Noble Gases
Noble gases (helium, neon, argon, krypton, xenon, and radon) are unique in their exceptional stability. They have a full outer electron shell (eight valence electrons, except for helium, which has two), making them extremely unreactive. Their lack of reactivity is why they're often referred to as "inert" gases. However, under specific conditions, some noble gases can form compounds.
Transition Metals: A Special Case
The transition metals occupy the central block of the periodic table (Groups 3-12). They differ from the main group elements in that their valence electrons are distributed across multiple electron shells, resulting in a more complex chemistry. Transition metals exhibit variable oxidation states, meaning they can lose different numbers of electrons to form ions with varying charges. This ability to adopt various oxidation states contributes to their diverse and often colorful compounds. Many transition metals are vital in biological systems, serving as essential components of enzymes and other biomolecules.
Trends Across Groups
As you move down a group in the periodic table, several trends are observed:
-
Atomic Radius: Atomic radius generally increases as you move down a group. This is because each successive element adds an additional electron shell, increasing the atom's size.
-
Electronegativity: Electronegativity, the ability of an atom to attract electrons in a chemical bond, generally decreases as you move down a group. As the atomic radius increases, the outermost electrons are farther from the nucleus, making them less strongly attracted.
-
Ionization Energy: Ionization energy, the energy required to remove an electron from an atom, generally decreases as you move down a group. The increased atomic radius means the outermost electrons are less tightly bound to the nucleus.
-
Reactivity: The reactivity trends vary depending on the group. For example, alkali metals become more reactive as you move down the group, while halogens become less reactive.
The Importance of Group Organization
The organization of the periodic table by groups is not merely a convenient arrangement; it provides invaluable insights into the behavior of elements. By understanding the characteristics of a particular group, chemists can predict the properties of its members and design experiments accordingly. This understanding is crucial in various fields, including:
-
Material Science: The properties of materials are intimately linked to the elements they contain. By choosing elements from specific groups, scientists can tailor the properties of materials for specific applications.
-
Medicine: Many elements are essential for biological processes, and understanding the properties of different groups helps in developing new drugs and therapies.
-
Environmental Science: Understanding the chemical behavior of elements helps in assessing environmental pollution and developing remediation strategies.
-
Industrial Chemistry: The chemical industry heavily relies on the properties of elements to produce a wide range of products, from plastics to fertilizers.
Conclusion: Groups - The Foundation of Chemical Understanding
The vertical columns of the periodic table, known as groups or families, represent a fundamental organizational principle reflecting the periodic recurrence of similar chemical properties. This similarity arises from the shared number of valence electrons, which dictate how elements interact and bond. Understanding group characteristics, trends, and the significance of valence electrons provides an essential foundation for comprehending the vast and complex world of chemistry. This knowledge forms the bedrock for advancements in diverse scientific and technological fields, highlighting the crucial role of group organization in unlocking the secrets of the elements and their interactions. The periodic table, with its elegantly organized groups and periods, remains a powerful tool that continues to shape our understanding of matter and the universe.
Latest Posts
Latest Posts
-
Which Of The Following Is Not Matched Correctly
Apr 03, 2025
-
A Ruptured Follicle Is Transformed Into The
Apr 03, 2025
-
List Some Properties For Ionic Compounds
Apr 03, 2025
-
How Many Degrees In A Parallelogram
Apr 03, 2025
-
4 Kingdoms In The Domain Eukarya
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Vertical Columns Of The Periodic Table Are Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
