Period 1 Contains A Total Of Elements
Juapaving
Apr 02, 2025 · 6 min read
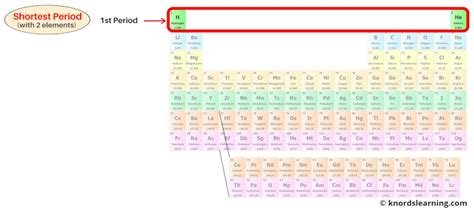
Table of Contents
Period 1: A Comprehensive Exploration of its Total Elemental Composition
The concept of "Period 1" most commonly refers to the first row of the periodic table of elements, containing only hydrogen (H) and helium (He). While seemingly simple, this period holds immense significance in understanding the fundamental building blocks of matter and the evolution of the universe. This article delves deeply into the properties, characteristics, and overall importance of these two elements, exploring their individual features and their combined role in shaping the cosmos.
Hydrogen: The Simplest and Most Abundant Element
Hydrogen, symbolized by H, is the lightest element in the periodic table, possessing only one proton and one electron in its neutral state. Its simplicity belies its profound importance. It's estimated to constitute approximately 75% of the universe's baryonic mass, making it the most abundant element.
Key Properties and Characteristics of Hydrogen:
-
Isotopes: Hydrogen boasts three naturally occurring isotopes: protium (¹H), deuterium (²H or D), and tritium (³H or T). These isotopes differ in the number of neutrons present in their nuclei. Protium, lacking a neutron, is the most common form. Deuterium, with one neutron, and tritium, with two neutrons, are heavier and less abundant.
-
Reactivity: Hydrogen is highly reactive, readily forming covalent bonds with many other elements. This reactivity stems from its single electron, which can be easily shared or transferred to achieve a stable electron configuration.
-
Oxidizing and Reducing Agent: Depending on the reaction, hydrogen can act as both an oxidizing agent (gaining electrons) and a reducing agent (losing electrons). This dual nature contributes to its versatility in chemical processes.
-
States of Matter: Hydrogen can exist in three states of matter: gaseous, liquid, and solid. Under standard conditions, it's a colorless, odorless, and tasteless diatomic gas (H₂).
-
Applications: Hydrogen finds wide-ranging applications, including:
- Ammonia Production (Haber-Bosch Process): A crucial component in the industrial synthesis of ammonia, a vital fertilizer.
- Fuel: A potential clean fuel source in fuel cells, producing only water as a byproduct.
- Metal Refining: Used in the refining of various metals.
- Chemical Synthesis: A fundamental reactant in countless chemical processes.
Hydrogen's Role in the Universe:
Hydrogen's prevalence in the universe is directly linked to its formation in the Big Bang. It's the primary component of stars, where nuclear fusion converts hydrogen into helium, releasing vast amounts of energy. This process powers the sun and other stars, providing light and warmth to planets and shaping the evolution of galaxies.
Helium: The Inert Noble Gas
Helium, symbolized by He, is the second-lightest and second-most abundant element in the universe. Unlike hydrogen, it's a noble gas, meaning it's chemically inert and rarely forms compounds. Its full valence shell gives it exceptional stability.
Key Properties and Characteristics of Helium:
-
Inertness: Helium's inertness is due to its complete electron shell, rendering it highly unreactive with other elements.
-
Low Density: Helium is the least dense gas after hydrogen, making it lighter than air. This property explains its use in balloons and airships.
-
Low Boiling Point: Helium possesses the lowest boiling point of all elements, remaining liquid even at extremely low temperatures. This characteristic makes it invaluable in cryogenics.
-
Superfluidity: At extremely low temperatures, helium exhibits superfluidity, a state of matter with zero viscosity, meaning it can flow without any resistance.
-
Isotopes: Like hydrogen, helium has several isotopes, with helium-4 (⁴He) being the most prevalent. Helium-3 (³He) is rarer and has potential applications in nuclear fusion research.
Helium's Significance and Applications:
-
Cryogenics: Helium's exceptionally low boiling point makes it indispensable in cryogenics, the science of producing and maintaining very low temperatures. It's used to cool superconducting magnets in MRI machines and other scientific instruments.
-
Welding: Helium's inert nature protects the weld zone from atmospheric contamination, improving the quality of welds.
-
Balloons and Airships: Its low density makes helium an ideal lifting gas for balloons and airships.
-
Leak Detection: Helium's ability to penetrate tiny leaks makes it useful in detecting leaks in high-vacuum systems.
-
Breathing Gas: Helium-oxygen mixtures are used by divers at great depths to reduce the risk of decompression sickness.
The Interplay Between Hydrogen and Helium: A Cosmic Dance
The relationship between hydrogen and helium is inextricably linked to the universe's evolution. The Big Bang nucleosynthesis produced vast quantities of hydrogen and helium, setting the stage for the formation of stars and galaxies. Within stars, hydrogen undergoes nuclear fusion to produce helium, releasing enormous energy in the process. This fusion process is the driving force behind stellar evolution and the generation of heavier elements.
The abundance of hydrogen and helium in the universe directly influences the formation of planets and planetary systems. The composition of gas giants like Jupiter and Saturn reflects the primordial abundance of these two elements. Furthermore, the interaction between hydrogen and helium plays a significant role in determining the physical and chemical properties of interstellar clouds and nebulae, the birthplaces of new stars and planetary systems.
Period 1's Influence on Subsequent Periods
While Period 1 contains only two elements, its influence on the subsequent periods of the periodic table is undeniable. The electronic structure of hydrogen and helium establishes the foundational principles of atomic structure and chemical bonding. The filling of electron shells, starting with hydrogen's single electron and progressing to helium's complete shell, lays the groundwork for understanding the periodic trends in electronegativity, ionization energy, and atomic radius observed in later periods.
The nuclear fusion reactions occurring within stars, transforming hydrogen into helium, generate the energy that drives stellar evolution and the creation of heavier elements. These heavier elements, formed through successive nuclear fusion reactions, populate the subsequent periods of the periodic table, enriching the chemical diversity of the universe.
Exploring Period 1 Beyond the Basics: Research and Future Applications
While the properties of hydrogen and helium are well understood, research continues to uncover new facets of these elements and their potential applications. For instance, research into hydrogen's use as a clean fuel source is progressing, focusing on efficient and safe storage and transportation methods. The potential of hydrogen fuel cells to power vehicles and other technologies remains a significant area of research and development.
Helium research is also ongoing, exploring its unique properties at extremely low temperatures and its potential applications in advanced technologies such as quantum computing. The quest for efficient ways to conserve and manage helium resources is also crucial, given its limited availability and increasing demand.
Conclusion: Period 1 – A Foundation for Understanding the Universe
In conclusion, Period 1, though containing only two elements, plays a pivotal role in understanding the universe's composition, evolution, and the fundamental principles of chemistry. Hydrogen and helium, the simplest and most abundant elements, are the building blocks of matter and the driving force behind stellar processes. Their unique properties and diverse applications continue to shape scientific advancements and technological innovations, highlighting their enduring importance in the world around us. Ongoing research continues to unlock new possibilities, cementing Period 1's enduring significance in our understanding of the cosmos and its constituents.
Latest Posts
Latest Posts
-
Balaneced Chemical Reaction Between C4h10 Oz Yeliding Co2
Apr 03, 2025
-
Which Is The Longest Phase In Meiosis
Apr 03, 2025
-
How Many Commas In A Billion
Apr 03, 2025
-
Glycolysis Ends In The Production Of
Apr 03, 2025
-
Distributive Law 3 Variables Discrete Math
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Period 1 Contains A Total Of Elements . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
