Mass Of A Proton In Grams
Juapaving
Apr 03, 2025 · 5 min read
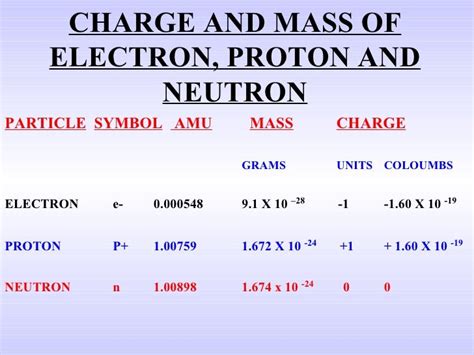
Table of Contents
The Mass of a Proton in Grams: A Deep Dive into Particle Physics
The proton, a fundamental building block of matter, holds a pivotal place in the realm of physics. Understanding its properties, especially its mass, is crucial to comprehending the universe's structure and behavior. While often expressed in atomic mass units (amu) or MeV/c², the proton's mass is frequently sought in grams. This article delves into the intricacies of determining and understanding the proton's mass in grams, exploring the underlying physics, conversion methods, and implications of this seemingly simple value.
Understanding the Proton's Mass
The mass of a proton is incredibly small, highlighting the minuscule scale of the subatomic world. It's not easily measured directly; rather, sophisticated experimental techniques and theoretical calculations are employed. The accepted value for the proton's mass is approximately 1.67262 × 10⁻²⁴ grams. However, this seemingly precise number is the result of extensive research and represents a weighted average of different proton states.
From Atomic Mass Units to Grams
The proton's mass is often initially expressed in atomic mass units (amu), where 1 amu is defined as one-twelfth the mass of a carbon-12 atom. This simplifies comparisons between different atomic and subatomic particles. The conversion from amu to grams relies on the precisely determined mass of a carbon-12 atom in grams:
- 1 amu ≈ 1.66054 × 10⁻²⁴ grams
Knowing that the proton's mass is approximately 1.007276 amu, we can perform the conversion:
1.007276 amu * (1.66054 × 10⁻²⁴ grams/amu) ≈ 1.67262 × 10⁻²⁴ grams
This simple conversion underscores the relationship between different units used in describing the mass of fundamental particles.
Methods for Determining Proton Mass
Accurately determining the proton's mass involves complex experimental procedures and theoretical models. Some of the key approaches include:
1. Mass Spectrometry:
Mass spectrometry is a powerful technique that measures the mass-to-charge ratio of ions. By ionizing protons and analyzing their trajectory in a magnetic field, researchers can precisely determine their mass. Advanced mass spectrometry techniques offer exceptional accuracy, contributing significantly to the refined value of the proton's mass. Different isotopes of hydrogen can also be analyzed and help in deriving a precise average proton mass.
2. Penning Traps:
Penning traps use a combination of electric and magnetic fields to confine charged particles. By measuring the particles' cyclotron frequency (the frequency of their circular motion in the magnetic field), researchers can determine their mass with extremely high precision. Penning traps are particularly useful for measuring the masses of fundamental particles with exceptionally high accuracy. The technique minimizes uncertainties and provides highly accurate measurements.
3. Quantum Electrodynamics (QED):
QED is a highly successful quantum field theory that describes the interaction between light and matter. Through complex calculations and incorporating experimental data, QED predictions of the proton's properties, including its mass, are made. The theoretical calculations are incredibly intricate, requiring advanced computational techniques and a deep understanding of fundamental interactions. QED provides essential theoretical support for the experimentally derived mass.
The Significance of the Proton's Mass
The proton's mass, seemingly insignificant on a macroscopic scale, has profound implications for various aspects of physics and cosmology:
1. Nuclear Physics:
The proton's mass is critical in understanding nuclear structure and stability. The strong nuclear force, responsible for binding protons and neutrons within atomic nuclei, is intimately linked to the masses of these particles. The mass defect (the difference between the mass of a nucleus and the sum of its constituent protons and neutrons) reveals the energy binding the nucleus together, and is related to the proton mass.
2. Particle Physics:
The proton's mass, alongside other fundamental particle masses, plays a central role in the Standard Model of particle physics. This model attempts to explain the fundamental forces and particles that make up the universe. However, the Standard Model does not fully explain the mass of the proton, requiring further theoretical investigation. The proton's mass is tightly coupled with the theory of the Higgs field, explaining how particles acquire their masses.
3. Cosmology:
The abundance of protons in the universe is a key factor in cosmological models. The early universe's conditions, including the relative abundance of protons and other particles, are essential in understanding the formation of galaxies and large-scale cosmic structures. The proton-to-neutron ratio in the early universe is directly influenced by the proton's mass.
4. Chemistry:
In chemistry, the proton's mass influences chemical reactions and properties. Isotope effects, which are differences in chemical behavior arising from variations in the number of neutrons within an atom, are directly related to the differences in mass between isotopes. The mass of the proton contributes to the overall mass of an atom.
The Uncertainty and Future Research
While the currently accepted value of the proton's mass in grams is remarkably precise, ongoing research continues to refine this value. The uncertainty associated with the measurement stems from the inherent complexities in experimental techniques and the limitations of theoretical models. Ongoing experiments with increasingly sophisticated equipment strive to reduce this uncertainty further. Future research into the proton's structure and the underlying mechanisms governing its mass will potentially unlock a deeper understanding of fundamental physics.
Conclusion
Determining the mass of a proton in grams, while seemingly a simple task, involves a remarkable blend of experimental ingenuity and theoretical sophistication. From mass spectrometry and Penning traps to the intricate calculations of QED, diverse approaches contribute to our understanding of this fundamental particle. The proton's mass is not merely a numerical value; it is a cornerstone of our understanding of the universe, with profound implications across various fields of physics and beyond. Continued research in this area promises further refinements and potentially revolutionary insights into the fundamental building blocks of reality. The quest for ever-greater precision in determining the proton's mass underscores the relentless pursuit of knowledge at the heart of scientific inquiry.
Latest Posts
Latest Posts
-
What Is The Lowest Common Multiple Of 8 And 12
Apr 04, 2025
-
Data And Information Are Interchangeable Terms True Or False
Apr 04, 2025
-
What Is The Least Common Multiple Of 24 And 16
Apr 04, 2025
-
How Many Feet Is 400 Yards
Apr 04, 2025
-
What Would Be The Major Product Of The Following Reaction
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Mass Of A Proton In Grams . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
