Is Pure Water A Mixture Or Compound
Juapaving
Apr 02, 2025 · 6 min read
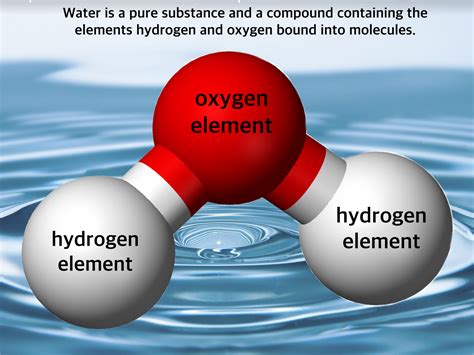
Table of Contents
Is Pure Water a Mixture or a Compound? A Deep Dive into the Chemistry of H₂O
The question of whether pure water is a mixture or a compound is a fundamental one in chemistry, often encountered early in scientific education. While the answer might seem simple at first glance, a deeper understanding requires exploring the definitions of mixtures and compounds, and the unique properties of water itself. This comprehensive guide will delve into the chemical composition of water, differentiate between mixtures and compounds, and definitively answer the question while exploring related concepts.
Understanding Mixtures and Compounds
Before we can classify pure water, we must clearly define the terms "mixture" and "compound." These represent distinct ways in which substances can combine:
Mixtures
A mixture is a substance comprising two or more components not chemically bonded. The components retain their individual chemical properties and can be separated using physical methods like filtration, distillation, evaporation, or chromatography. Mixtures can be homogeneous (uniform composition throughout, like saltwater) or heterogeneous (non-uniform composition, like sand and water). The proportions of the components in a mixture can vary.
Examples of Mixtures:
- Air: A mixture of gases, primarily nitrogen, oxygen, and argon.
- Saltwater: A homogeneous mixture of salt (sodium chloride) dissolved in water.
- Soil: A heterogeneous mixture of minerals, organic matter, and water.
Compounds
A compound, on the other hand, is a substance formed when two or more chemical elements are chemically bonded together. These bonds involve the sharing or transfer of electrons, resulting in a new substance with properties different from its constituent elements. Compounds have a fixed composition; their elements are always present in a specific ratio by mass. Separating the elements of a compound requires chemical reactions, not physical methods.
Examples of Compounds:
- Water (H₂O): Composed of two hydrogen atoms and one oxygen atom, covalently bonded.
- Sodium chloride (NaCl): Composed of sodium and chlorine atoms, ionically bonded.
- Carbon dioxide (CO₂): Composed of one carbon atom and two oxygen atoms, covalently bonded.
The Chemical Structure of Water
Water, with its chemical formula H₂O, is composed of two hydrogen atoms covalently bonded to a single oxygen atom. This covalent bond involves the sharing of electrons between the oxygen and hydrogen atoms, resulting in a stable molecule. The oxygen atom is more electronegative than the hydrogen atoms, meaning it attracts the shared electrons more strongly. This creates a polar molecule, with a slightly negative charge on the oxygen atom and slightly positive charges on the hydrogen atoms. This polarity is crucial to many of water's unique properties.
The Covalent Bond in Water: A Closer Look
The covalent bond in water is a strong chemical bond, requiring significant energy to break. This strength is a key factor in water's stability and its role as a solvent. The bond is formed through the overlapping of atomic orbitals, creating a shared electron pair between the oxygen and each hydrogen atom. The electron pair is not shared equally, resulting in the polar nature of the water molecule.
Why Pure Water is a Compound, Not a Mixture
Given the definitions of mixtures and compounds, it's clear that pure water is a compound, not a mixture. This is because:
-
Fixed Composition: Pure water always contains two hydrogen atoms and one oxygen atom in a fixed ratio (2:1 by atom count, approximately 11.19% hydrogen and 88.81% oxygen by mass). This fixed ratio is characteristic of compounds, not mixtures. You cannot change this ratio and still have pure water.
-
Chemical Bonding: The hydrogen and oxygen atoms in water are chemically bonded through strong covalent bonds. These bonds hold the atoms together as a single unit, unlike in mixtures where components are simply physically intermingled.
-
Unique Properties: Water possesses unique properties that differ significantly from its constituent elements, hydrogen and oxygen. Hydrogen is a highly flammable gas, while oxygen is a vital component of combustion. Water, however, is neither flammable nor does it readily support combustion. These differing properties are a direct result of the chemical bonding within the water molecule. Mixtures, on the other hand, retain the properties of their constituent components.
-
Chemical Decomposition: To separate water into its constituent elements (hydrogen and oxygen), a chemical reaction is required, such as electrolysis. This contrasts with mixtures, where physical methods suffice for separation.
Addressing Common Misconceptions
Some might argue that water found in nature is never truly "pure," containing dissolved minerals and other impurities. While this is true, the classification of water as a compound or mixture depends on its purity. Pure water, free from any other substance, is a compound. Natural water, with dissolved minerals, is a mixture of water (the compound) and those dissolved substances. The distinction is crucial.
The Importance of Pure Water
The concept of pure water holds immense significance in various fields:
-
Scientific Research: Pure water is essential in numerous scientific experiments and analytical techniques, providing a controlled environment free from interfering substances.
-
Pharmaceutical Industry: Pure water is crucial for the production of pharmaceuticals, ensuring product purity and safety.
-
Industrial Applications: Many industrial processes require pure water to avoid contamination and maintain product quality.
-
Human Consumption: While we don't consume perfectly pure water (due to the practical difficulties of achieving this), understanding the purity requirements for potable water is crucial for public health and safety.
Water's Unique Properties: A Consequence of its Chemical Structure
The unique properties of water, often dubbed "the universal solvent," arise directly from its chemical structure and the polarity of its molecules:
-
High Specific Heat Capacity: Water can absorb a large amount of heat without a significant temperature change. This property moderates Earth's temperature and plays a vital role in regulating the climate.
-
High Heat of Vaporization: A significant amount of energy is required to change water from liquid to gas. This property facilitates cooling through evaporation.
-
Excellent Solvent: Water's polarity allows it to dissolve many ionic and polar compounds, making it an excellent solvent for numerous biological and chemical processes.
-
High Surface Tension: Water molecules exhibit strong cohesive forces, resulting in high surface tension. This property is essential for capillary action in plants and many other biological phenomena.
-
Density Anomaly: Ice is less dense than liquid water, a unique property that allows aquatic life to survive in colder climates.
Conclusion: Pure Water is a Compound
In conclusion, pure water is unequivocally a compound. Its fixed composition, strong chemical bonds, unique properties, and the requirement of chemical processes for decomposition all align with the definition of a compound, not a mixture. While natural water sources often contain impurities, making them mixtures, the fundamental building block – pure water – is a chemically bonded substance that exhibits properties distinctly different from its constituent elements. This understanding is critical for numerous scientific, industrial, and everyday applications. The fascinating properties of water stem directly from the unique chemical bonds within its simple H₂O molecule, underscoring the power and elegance of chemical structure.
Latest Posts
Latest Posts
-
Compare And Contrast Pulmonary Circulation With Systemic Circulation
Apr 03, 2025
-
Does Photosynthesis Take Place Primarily In Plant Leaves
Apr 03, 2025
-
A Sphere Is Inscribed In A Cube
Apr 03, 2025
-
Balanced Equation Of Hcl And Naoh
Apr 03, 2025
-
What Is 0 35 As A Fraction
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is Pure Water A Mixture Or Compound . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
