Is Baking Soda Acid Or Base
Juapaving
Apr 04, 2025 · 5 min read
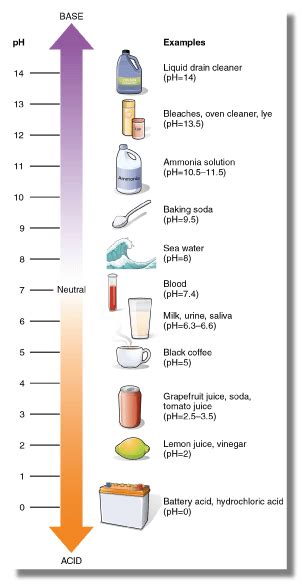
Table of Contents
Is Baking Soda Acid or Base? Understanding pH and its Culinary Applications
Baking soda, a staple in many kitchens, is often associated with baking, but its true nature lies in its chemical properties. The question, "Is baking soda acid or base?", is a fundamental one that unlocks a deeper understanding of its functionality and versatility. This comprehensive guide will delve into the chemical makeup of baking soda, explore its pH level, and examine its various applications, particularly in baking and beyond. We'll also look at how it interacts with other ingredients, clarifying common misconceptions.
Understanding pH: The Acid-Base Scale
Before we tackle the baking soda question, let's establish a clear understanding of the pH scale. The pH scale measures the acidity or alkalinity of a substance, ranging from 0 to 14. A pH of 7 is considered neutral (like pure water). Values below 7 indicate acidity, with lower numbers representing stronger acids (e.g., battery acid). Values above 7 indicate alkalinity or basicity, with higher numbers indicating stronger bases (e.g., lye).
Key Terms:
- Acid: A substance that donates hydrogen ions (H+) in a solution.
- Base: A substance that accepts hydrogen ions (H+) in a solution or donates hydroxide ions (OH-).
- pH: A measure of the hydrogen ion concentration in a solution.
Baking Soda: A Basic Compound
The answer is clear: baking soda is a base. Its chemical name is sodium bicarbonate (NaHCO₃). When dissolved in water, it releases bicarbonate ions (HCO₃⁻), which act as weak bases. These bicarbonate ions can accept hydrogen ions, thus increasing the solution's pH.
The Chemical Reaction: Understanding Bicarbonate's Behavior
The basicity of baking soda stems from the bicarbonate ion's ability to react with acids. This reaction is crucial to its leavening properties in baking. The bicarbonate ion readily accepts a proton (H+) from an acid, forming carbonic acid (H₂CO₃). Carbonic acid is unstable and quickly decomposes into water (H₂O) and carbon dioxide (CO₂). This CO₂ gas is what creates the rise in baked goods.
The Reaction:
NaHCO₃ + H⁺ → H₂CO₃ → H₂O + CO₂
This reaction highlights the crucial role of baking soda as a base: it needs an acid to react and produce the leavening effect. This is why baking soda is often used in combination with acidic ingredients.
Baking Soda in Baking: The Acid-Base Reaction in Action
Baking soda's function in baking is entirely dependent on its reaction with acids. Without an acid, baking soda remains inactive and won't produce the desired leavening effect. In fact, it can leave a soapy or bitter aftertaste.
Common Acidic Ingredients in Baking:
- Buttermilk: Contains lactic acid.
- Yogurt: Contains lactic acid.
- Lemon juice: Contains citric acid.
- Vinegar: Contains acetic acid.
- Brown sugar: Contains small amounts of organic acids.
- Molasses: Contains small amounts of organic acids.
- Honey: Contains small amounts of organic acids.
- Cocoa powder: Contains small amounts of organic acids.
The combination of baking soda and an acid creates a chemical reaction that produces carbon dioxide gas, causing the batter or dough to rise. The timing of this reaction is crucial for achieving the desired texture and volume in baked goods.
Baking Powder: A Pre-Mixed Solution
Baking powder contains both baking soda and an acid (usually cream of tartar). This pre-mixed combination ensures that the leavening reaction occurs even without the addition of acidic ingredients. However, baking powder often contains multiple types of acids for a two-stage leavening reaction, providing lift both initially and during the baking process.
Baking Soda Beyond Baking: Other Applications
The basicity of baking soda extends its utility far beyond the realm of baking. Its diverse applications showcase its remarkable versatility:
Cleaning and Deodorizing:
- Cleaning agent: Baking soda's mildly abrasive nature and its ability to neutralize acids make it an effective cleaning agent for various surfaces. It can be used to scrub pots and pans, clean countertops, and even brighten whites in laundry.
- Deodorizer: Baking soda is an excellent odor absorber. It can be placed in refrigerators, freezers, and other areas prone to unpleasant smells. Its ability to neutralize acids is key to this function.
Health and Beauty:
- Toothpaste ingredient: Baking soda's mild abrasiveness is used in some toothpastes to help remove surface stains. However, overuse can damage tooth enamel.
- Antacid: Baking soda's ability to neutralize stomach acid can provide temporary relief from heartburn. Consult a doctor for chronic issues.
- Exfoliant: Baking soda can be added to face washes or scrubs for gentle exfoliation. However, its use should be infrequent to avoid irritation.
Other Uses:
- Fire extinguisher: Baking soda can extinguish small grease fires by interrupting the combustion process.
- Gardening: Baking soda can help regulate soil pH, and it can be used as a fungicide for plants.
Common Misconceptions about Baking Soda
Several misconceptions surround baking soda's use, often stemming from a lack of understanding of its chemical properties:
Misconception 1: Baking Soda is only for cookies.
Reality: While frequently used in cookies, baking soda's applications extend far beyond cookies. Its ability to react with acids allows for use in cakes, breads, muffins, and other baked goods requiring a leavening agent.
Misconception 2: Baking soda can be used as a direct replacement for baking powder.
Reality: Baking soda and baking powder are not interchangeable. Baking soda requires an acid for activation, while baking powder contains its own acid component. Using only baking soda without an acid will result in a flat, unrisen product. Conversely, using baking powder when an acidic ingredient is already present might lead to excessive leavening.
Misconception 3: Baking soda is harmful.
Reality: In appropriate amounts and applications, baking soda is generally safe for consumption and use. However, excessive ingestion can cause health problems. Always follow instructions and use appropriate safety precautions, particularly when handling it as a cleaning agent.
Conclusion: A Versatile Base with Broad Applications
Baking soda, a seemingly simple ingredient, is in fact a potent base with a wide array of applications. Its ability to react with acids, producing carbon dioxide gas, is central to its role in baking. However, its functionality extends far beyond the kitchen, encompassing cleaning, deodorizing, and even some health and beauty uses. By understanding its chemical nature and properties, you can harness the versatility of this remarkable compound effectively and safely. Remember always to consult relevant resources and follow safety instructions when working with any chemical substance. Its multifaceted nature and readily available status cement its position as a true kitchen and household staple.
Latest Posts
Latest Posts
-
Words With An A And Z
Apr 04, 2025
-
Finding The Area And Perimeter Of A Triangle
Apr 04, 2025
-
How Many Thousands Is In A Million
Apr 04, 2025
-
Difference Between A State And Union Territory
Apr 04, 2025
-
5 Letter Words Start With As
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Is Baking Soda Acid Or Base . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
