How Many Neutrons Does A Hydrogen Atom Have
Juapaving
Apr 02, 2025 · 5 min read
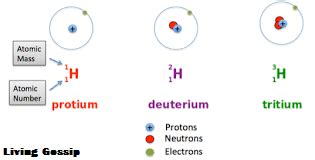
Table of Contents
How Many Neutrons Does a Hydrogen Atom Have? A Deep Dive into Isotopes
The seemingly simple question, "How many neutrons does a hydrogen atom have?" leads us down a fascinating path exploring the intricacies of atomic structure, isotopes, and the implications for various scientific fields. The short answer is: it depends. Unlike the number of protons which defines an element, the number of neutrons can vary, leading to different isotopes of the same element. Let's delve deeper into this topic.
Understanding Atomic Structure: Protons, Neutrons, and Electrons
Before we address the neutron count in hydrogen, it's crucial to understand the basic building blocks of an atom. Every atom consists of three primary subatomic particles:
- Protons: Positively charged particles located in the atom's nucleus. The number of protons determines the element's atomic number and its identity on the periodic table. Hydrogen, with an atomic number of 1, possesses one proton.
- Neutrons: Neutrally charged particles residing in the atom's nucleus alongside protons. Unlike protons, the number of neutrons can vary within the same element, leading to different isotopes.
- Electrons: Negatively charged particles orbiting the nucleus in electron shells. Electrons are significantly lighter than protons and neutrons and determine the atom's chemical properties and reactivity. In a neutral atom, the number of electrons equals the number of protons.
Isotopes of Hydrogen: A Closer Look
Hydrogen, the simplest element, has three naturally occurring isotopes, each differing in its neutron count:
1. Protium (¹H): The Most Abundant Isotope
Protium is the most common isotope of hydrogen, comprising about 99.98% of all naturally occurring hydrogen. As indicated by its symbol ¹H, it possesses:
- 1 proton
- 0 neutrons
- 1 electron
This is the simplest form of a hydrogen atom, with its nucleus containing only a single proton. Its simplicity makes it crucial in various chemical reactions and processes.
2. Deuterium (²H or D): A Stable Isotope with One Neutron
Deuterium, also known as heavy hydrogen, is a stable isotope of hydrogen. Its symbol, ²H or D, reflects its atomic mass, which is approximately twice that of protium. Deuterium contains:
- 1 proton
- 1 neutron
- 1 electron
The presence of an extra neutron significantly increases its mass, leading to slightly different physical and chemical properties compared to protium. While heavier, deuterium is stable and plays a role in various scientific applications, including nuclear magnetic resonance (NMR) spectroscopy and nuclear fusion research.
3. Tritium (³H or T): A Radioactive Isotope with Two Neutrons
Tritium, a radioactive isotope of hydrogen, is much less abundant than protium and deuterium. Its symbol, ³H or T, indicates its atomic mass. Tritium consists of:
- 1 proton
- 2 neutrons
- 1 electron
The presence of two neutrons makes tritium radioactive, undergoing beta decay with a half-life of approximately 12.3 years. This radioactive decay transforms a neutron into a proton, emitting a beta particle (an electron) and an antineutrino. Tritium finds applications in various fields, including nuclear fusion research, self-powered lighting devices, and as a tracer in biological and environmental studies. However, due to its radioactivity, its handling requires stringent safety precautions.
Implications of Different Neutron Counts
The varying neutron counts in hydrogen isotopes significantly affect their properties and applications. These differences are primarily manifested in:
- Mass: The additional neutrons in deuterium and tritium increase their mass compared to protium. This mass difference influences their physical properties, such as density and diffusion rates.
- Nuclear Stability: Tritium's two neutrons render it unstable and radioactive, unlike protium and deuterium, which are stable. This radioactivity has implications for its use and necessitates safety measures.
- Chemical Properties: While the chemical properties of hydrogen isotopes are largely similar due to their identical electron configurations, subtle differences exist due to the isotope effect, influencing reaction rates and equilibrium constants.
- Applications: The unique properties of each isotope dictate its specific applications. Protium finds use in numerous chemical processes, deuterium in NMR spectroscopy and fusion research, and tritium in specialized applications like self-powered lighting and tracers.
Hydrogen Isotopes in Various Fields
The distinct properties of hydrogen's isotopes make them indispensable tools across multiple scientific and technological fields:
- Nuclear Fusion: Deuterium and tritium are critical fuels in nuclear fusion reactions, aiming to replicate the energy production processes of stars. Their fusion releases immense energy, offering a potential clean energy source.
- Nuclear Magnetic Resonance (NMR) Spectroscopy: Deuterium's unique NMR properties make it valuable in analytical chemistry for identifying and analyzing molecules.
- Isotope Tracing: Tritium's radioactivity allows its use as a tracer to track the movement and behavior of molecules in biological and environmental systems.
- Medical Imaging: Deuterium oxide (heavy water) has found use in some medical imaging techniques.
- Material Science: Deuterium and tritium can be used to study materials' behavior under specific conditions.
Conclusion: The Variable Nature of Neutrons in Hydrogen
The number of neutrons in a hydrogen atom is not a fixed value; it varies depending on the isotope. Protium has zero neutrons, deuterium one, and tritium two. These variations directly affect the isotopes' physical and chemical properties, leading to a wide range of applications across various scientific and technological disciplines. Understanding these differences is critical for researchers and scientists working in fields ranging from nuclear physics and chemistry to biology and medicine. The seemingly simple question of neutron count in hydrogen underscores the complex and fascinating world of atomic structure and isotopic variation. The exploration of these variations opens doors to significant advancements in energy production, analytical techniques, and scientific understanding.
Latest Posts
Latest Posts
-
Choose The True Statement About The Krebs Cycle
Apr 03, 2025
-
Essay On The Pleasure Of Reading
Apr 03, 2025
-
Compare And Contrast Pulmonary Circulation With Systemic Circulation
Apr 03, 2025
-
Does Photosynthesis Take Place Primarily In Plant Leaves
Apr 03, 2025
-
A Sphere Is Inscribed In A Cube
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Neutrons Does A Hydrogen Atom Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
