How Many Neutrons Are In Potassium
Juapaving
Apr 02, 2025 · 4 min read
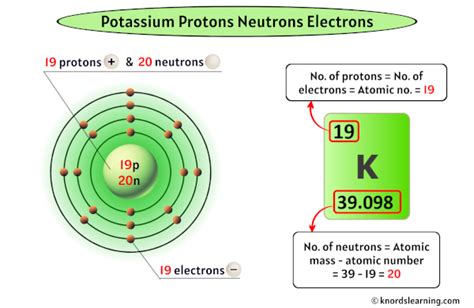
Table of Contents
How Many Neutrons Are in Potassium? A Deep Dive into Isotopes and Atomic Structure
Potassium, a vital element for human life and a common component in fertilizers, presents an interesting case study in atomic structure. While the periodic table tells us its atomic number (the number of protons), understanding the number of neutrons requires a closer look at isotopes. This article will delve into the complexities of potassium isotopes, explaining how to determine the neutron count and exploring the implications of different isotopic compositions.
Understanding Atomic Structure: Protons, Neutrons, and Electrons
Before we dive into potassium's neutrons, let's establish a foundation in basic atomic structure. An atom consists of three fundamental particles:
-
Protons: Positively charged particles found in the nucleus (the atom's core). The number of protons determines the element's atomic number and its identity on the periodic table. Potassium's atomic number is 19, meaning every potassium atom has 19 protons.
-
Neutrons: Neutrally charged particles also residing in the nucleus. Unlike protons, the number of neutrons can vary within the same element, leading to different isotopes.
-
Electrons: Negatively charged particles orbiting the nucleus in electron shells. They are significantly lighter than protons and neutrons and are involved in chemical bonding. In a neutral atom, the number of electrons equals the number of protons.
Isotopes: The Key to Understanding Neutron Numbers
The key to determining the number of neutrons in potassium lies in understanding isotopes. Isotopes are atoms of the same element (same number of protons) but with a different number of neutrons. This difference in neutron number results in variations in atomic mass.
Potassium has three naturally occurring isotopes:
-
Potassium-39 (³⁹K): This is the most abundant isotope, making up about 93.3% of naturally occurring potassium. It has 19 protons and 20 neutrons (39 - 19 = 20).
-
Potassium-40 (⁴⁰K): A radioactive isotope, potassium-40 accounts for about 0.012% of natural potassium. It has 19 protons and 21 neutrons (40 - 19 = 21). This isotope is important in geological dating and has implications for radiation exposure.
-
Potassium-41 (⁴¹K): This is the second most abundant isotope, composing roughly 6.7% of natural potassium. It contains 19 protons and 22 neutrons (41 - 19 = 22).
Calculating the Number of Neutrons: A Simple Formula
The number of neutrons in an atom can be easily calculated using the following formula:
Number of neutrons = Mass number - Atomic number
Where:
- Mass number: The total number of protons and neutrons in an atom's nucleus (represented by the superscript number before the element symbol, e.g., ³⁹K).
- Atomic number: The number of protons in the atom's nucleus (this is the element's position on the periodic table).
Let's apply this formula to the three potassium isotopes:
- ³⁹K: 39 - 19 = 20 neutrons
- ⁴⁰K: 40 - 19 = 21 neutrons
- ⁴¹K: 41 - 19 = 22 neutrons
The Significance of Potassium Isotopes
The varying numbers of neutrons in potassium isotopes have several significant consequences:
Radioactive Decay of Potassium-40
Potassium-40 is a radioactive isotope, undergoing beta decay. This means it emits a beta particle (an electron) and transforms into Calcium-40. This decay process is crucial for:
-
Geological dating: The decay rate of potassium-40 is constant, allowing scientists to date rocks and minerals accurately. By measuring the ratio of potassium-40 to argon-40 (its decay product), geologists can estimate the age of geological formations.
-
Radiation exposure: While the amount of potassium-40 in our bodies is small, its radioactive decay contributes to a small but measurable dose of background radiation.
Biological Roles and Isotopic Abundance
The different isotopes of potassium play varying roles in biological systems. While the body does not differentiate between the isotopes in terms of chemical behavior, the different isotopic abundances have implications for research in various fields:
-
Metabolic studies: Scientists utilize stable isotopes like potassium-39 and potassium-41 as tracers to study potassium metabolism in organisms.
-
Nutrient uptake studies: Isotopic analysis can reveal details about nutrient uptake and transport in plants. The relative abundances of potassium isotopes in plants can provide insights into their growth conditions and nutrient sources.
Beyond Natural Potassium: Artificial Isotopes
While naturally occurring potassium consists primarily of three isotopes, scientists have also created various artificial potassium isotopes through nuclear reactions. These isotopes are typically radioactive and have short half-lives, meaning they decay quickly into other elements. These artificial isotopes have uses in various scientific applications, particularly in nuclear medicine and research.
Conclusion: A Deeper Understanding of Potassium
Understanding the number of neutrons in potassium requires appreciating the concept of isotopes. The three naturally occurring isotopes – potassium-39, potassium-40, and potassium-41 – each possess a different neutron count, contributing to their unique properties and roles in various contexts, from geological dating to biological processes. This knowledge highlights the importance of considering isotopic variations when discussing elements and their properties and emphasizes the fundamental relationship between atomic structure and its macroscopic consequences. The relatively simple calculation of neutrons based on mass and atomic numbers allows for deeper comprehension of the complexities inherent in the atomic world. Further investigation into these intricacies of potassium and its isotopes can lead to advancements in various scientific fields.
Latest Posts
Latest Posts
-
Which Of The Following Is Not A Compound
Apr 03, 2025
-
What Is The Lcm Of 3 And 7
Apr 03, 2025
-
A Circle Is Continuously Rotated About Its Diameter
Apr 03, 2025
-
How Many Feet In 25 Meters
Apr 03, 2025
-
What Is The Most Common Gas In The Atmosphere
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Neutrons Are In Potassium . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
