How Many Electrons Does A Sulfur Atom Have
Juapaving
Apr 02, 2025 · 5 min read
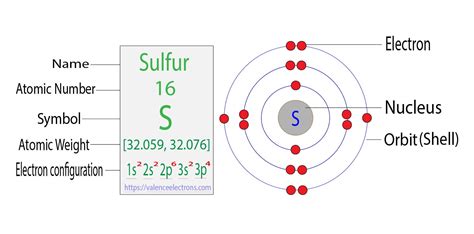
Table of Contents
How Many Electrons Does a Sulfur Atom Have? A Deep Dive into Atomic Structure
Understanding the number of electrons in a sulfur atom is fundamental to grasping its chemical properties and behavior. This seemingly simple question opens the door to a fascinating exploration of atomic structure, electron configuration, and the periodic table. Let's delve into the intricacies of sulfur's electron count and explore its implications.
Understanding Atomic Structure: Protons, Neutrons, and Electrons
Before we pinpoint the electron count of sulfur, let's review the basic building blocks of an atom:
- Protons: Positively charged particles found in the atom's nucleus. The number of protons defines the element; it's the atomic number.
- Neutrons: Neutrally charged particles also residing in the nucleus. The number of neutrons can vary within an element, leading to isotopes.
- Electrons: Negatively charged particles orbiting the nucleus in energy levels or shells. The number of electrons typically equals the number of protons in a neutral atom.
This balance of positive and negative charges is what makes a neutral atom. If an atom loses or gains electrons, it becomes an ion, carrying a net positive (cation) or negative (anion) charge.
Finding Sulfur on the Periodic Table
The periodic table is a chemist's best friend. It organizes elements based on their atomic number and recurring chemical properties. Locating sulfur on the periodic table is crucial for determining its electron count.
Sulfur (S) is found in the third period (row) and the 16th group (column), also known as Group VIA or Group 16. Its atomic number is 16. This atomic number directly tells us the number of protons in a sulfur atom's nucleus.
The Crucial Connection: Atomic Number and Electron Count
Because a neutral atom has an equal number of protons and electrons, the atomic number provides the answer to our question. Since sulfur's atomic number is 16, a neutral sulfur atom has 16 electrons.
Electron Shells and Subshells: A Deeper Look at Electron Configuration
Electrons don't just randomly orbit the nucleus. They occupy specific energy levels called shells, and within those shells, subshells. Understanding this electron configuration helps explain sulfur's chemical reactivity.
The electron configuration of sulfur is 1s²2s²2p⁶3s²3p⁴. Let's break this down:
- 1s²: The first shell (n=1) contains one subshell (s), holding a maximum of two electrons. Sulfur has two electrons in this lowest energy level.
- 2s²: The second shell (n=2) also has an s subshell, accommodating another two electrons.
- 2p⁶: The second shell also has three p subshells (px, py, pz), each capable of holding two electrons, for a total of six electrons in this subshell.
- 3s²: The third shell (n=3) begins with an s subshell containing two electrons.
- 3p⁴: Finally, the third shell's three p subshells contain four electrons. This is where sulfur's reactivity stems from – it needs four more electrons to fill its outermost shell completely.
Sulfur's Valence Electrons: The Key to Reactivity
The outermost shell of an atom, containing the valence electrons, determines its chemical behavior. Sulfur's valence electrons are the four electrons in the 3p subshell. These electrons are readily involved in forming chemical bonds with other atoms. This tendency to gain or share electrons to achieve a stable octet (eight electrons in the outer shell) makes sulfur highly reactive.
Isotopes of Sulfur: Variations in Neutron Count
While the number of electrons typically matches the number of protons, the number of neutrons can vary. These variations create isotopes of sulfur. All isotopes of sulfur have 16 protons, but the neutron count differs. Common isotopes include sulfur-32, sulfur-33, sulfur-34, and sulfur-36. The number after the element's name represents the mass number (protons + neutrons). The electron count remains 16 for all neutral isotopes of sulfur.
Sulfur's Role in Chemical Reactions: Putting the Electrons to Work
The 16 electrons in a sulfur atom play a crucial role in its chemical behavior. Its tendency to gain two electrons to complete its octet makes it readily form anions with a -2 charge (S²⁻). This explains its presence in numerous ionic compounds like sulfides. It also readily forms covalent bonds by sharing electrons with other atoms, as seen in molecules like sulfur dioxide (SO₂) and sulfuric acid (H₂SO₄).
Sulfur's Importance in Biological Systems: A Vital Element
Sulfur is an essential element for life. It's a component of amino acids, proteins, and enzymes, playing a pivotal role in numerous biological processes. Its presence in vital molecules underscores the significance of its electron configuration and reactivity.
Applications of Sulfur and its Compounds: From Industry to Medicine
Sulfur's properties and the unique arrangement of its 16 electrons lead to its extensive applications:
- Vulcanization of rubber: Sulfur is crucial in improving the elasticity and durability of rubber, a process essential for tire manufacturing.
- Production of sulfuric acid: Sulfuric acid, a highly corrosive compound, is a cornerstone of many industrial processes, including fertilizer production and metal refining.
- Pharmaceuticals: Sulfur compounds find applications in medications, including some antifungals and antibacterials.
- Matches and fireworks: Sulfur's flammability contributes to its use in pyrotechnics.
- Fertilizers: Sulfur is an essential nutrient for plant growth, and sulfur-containing fertilizers are vital for agriculture.
Conclusion: The Significance of Sulfur's 16 Electrons
The seemingly simple answer – a sulfur atom has 16 electrons – opens a window into the fascinating world of atomic structure, chemical bonding, and the remarkable properties that emerge from the precise arrangement of these subatomic particles. The number 16 isn't just a number; it's a key that unlocks understanding of sulfur's behavior, its importance in the natural world, and its widespread applications in various technologies and industries. From its role in biological systems to its industrial applications, sulfur's 16 electrons are fundamental to its vital functions. Understanding this fundamental aspect is essential for appreciating the remarkable influence of this element on our world.
Latest Posts
Latest Posts
-
How To Calculate The Perimeter Of A Rhombus
Apr 03, 2025
-
Is The Electromagnetic Spectrum Transverse Or Longitudinal
Apr 03, 2025
-
Compare And Contrast Pulmonary And Systemic Circulation
Apr 03, 2025
-
Icse Tuitions Classes In Jail Road Nashik
Apr 03, 2025
-
What Is A Free Variable In A Matrix
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Electrons Does A Sulfur Atom Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
