Explain Why The Noble Gases Are Unreactive
Juapaving
Apr 04, 2025 · 5 min read
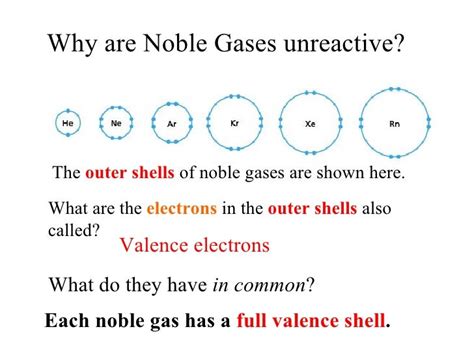
Table of Contents
Why Are Noble Gases Unreactive? Unraveling the Mystery of Inertness
The noble gases, also known as inert gases, occupy Group 18 of the periodic table. Their unique characteristic, and the reason for their name, is their exceptional chemical inertness. Unlike most other elements that readily react to form compounds, noble gases stubbornly resist forming chemical bonds. This remarkable unreactivity stems from their electronic structure, specifically their complete valence electron shells. This article will delve deep into the reasons behind this inertness, exploring the quantum mechanical principles and exploring some exceptions to this general rule.
The Octet Rule: A Foundation of Chemical Stability
The key to understanding the unreactivity of noble gases lies in the octet rule. This rule states that atoms tend to gain, lose, or share electrons in order to achieve a full outer electron shell, typically containing eight electrons (except for hydrogen and helium). This stable configuration resembles the electron arrangement of noble gases, conferring significant stability. Noble gases already possess this stable configuration naturally, meaning they have little to no incentive to participate in chemical reactions.
Helium's Special Case: The Duet Rule
Helium, the lightest noble gas, deviates slightly from the octet rule. Its outermost shell can only accommodate two electrons, and it already possesses this complete duet. This full shell explains helium's exceptional stability and inertness.
Valence Electrons and Electron Configurations: The Key to Inertness
The noble gases' electronic structures are characterized by completely filled valence electron shells. Valence electrons are the electrons in the outermost shell of an atom; these electrons are most directly involved in chemical bonding. A filled valence shell signifies maximum stability.
Let's examine the electron configurations of some noble gases:
- Helium (He): 1s² (2 electrons)
- Neon (Ne): 1s²2s²2p⁶ (10 electrons)
- Argon (Ar): 1s²2s²2p⁶3s²3p⁶ (18 electrons)
- Krypton (Kr): 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁶ (36 electrons)
- Xenon (Xe): 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁶5s²4d¹⁰5p⁶ (54 electrons)
Notice how each element has a completely filled outermost shell. This complete shell requires a significant amount of energy to disrupt, making them incredibly resistant to forming chemical bonds.
Ionization Energy and Electron Affinity: High Energy Barriers
The reluctance of noble gases to react is also reflected in their high ionization energy and low electron affinity.
- Ionization energy is the energy required to remove an electron from an atom. For noble gases, this energy is very high because removing an electron would disrupt their stable, filled electron shell.
- Electron affinity is the energy change that occurs when an atom gains an electron. Noble gases have low electron affinities, meaning that adding an electron is not energetically favorable; they don't want to disrupt their already stable state.
Why Reaction is Unfavorable: A Thermodynamic Perspective
From a thermodynamic standpoint, chemical reactions proceed spontaneously only if they are accompanied by a decrease in Gibbs Free Energy (ΔG). For noble gases to react, they would need to overcome the significant energy barrier represented by their high ionization energy and the lack of energy gain from electron affinity. This makes the ΔG for reactions involving noble gases highly positive, thus rendering them thermodynamically unfavorable.
Moreover, the strong interatomic forces within the noble gas atoms due to their stable electron configurations further prevent the formation of chemical bonds. The energy required to overcome these forces and create new bonds is usually far greater than any potential energy released during the bonding process.
Exceptions to the Rule: The Rare Compounds of Xenon
While the overwhelming majority of evidence supports the inert nature of noble gases, a few exceptions exist. Under extreme conditions (high pressures, low temperatures, and special catalysts), xenon, the most reactive of the noble gases, can form compounds, primarily with highly electronegative elements like fluorine and oxygen. Examples include xenon difluoride (XeF₂) and xenon tetrafluoride (XeF₄).
These compounds challenge the historical classification of noble gases as completely unreactive but do not invalidate the general principle. The formation of these compounds can be attributed to the high electronegativity of fluorine, which can polarize the electron cloud of xenon, thus weakening the stability of its filled shell to a certain extent under specific conditions. The larger size of xenon also allows for the possibility of a greater number of interactions, further influencing its limited reactivity.
Applications of Noble Gases: Leveraging Their Inertness
The unreactivity of noble gases is precisely what makes them so useful in various applications. Their inertness makes them ideal for scenarios where preventing reactions is critical:
- Arc welding: Noble gases, such as argon, provide an inert atmosphere to protect the weld from oxidation.
- Lighting: Neon, argon, krypton, and xenon are used in various types of lighting, taking advantage of their ability to emit light when an electric current passes through them.
- Cryogenics: Helium's extremely low boiling point (-268.93 °C) makes it crucial in cryogenics, the study of extremely low temperatures, including MRI machines and superconducting magnets.
- Medical applications: Helium and argon find applications in various medical procedures.
- Laser technology: Noble gases play a role in producing specific types of lasers.
Conclusion: Inertness, but not Inactivity
The noble gases' unreactivity is a consequence of their complete valence electron shells, high ionization energies, and low electron affinities. This exceptional stability makes them unique among the elements and dictates their numerous applications where inertness is paramount. While the existence of a small number of xenon compounds demonstrates that their inertness isn't absolute, the overall principle of their exceptional stability and resistance to chemical reactions remains firmly established. Their exceptional properties continue to be exploited across numerous scientific and technological fields, solidifying their importance in our modern world. The study of these elements provides a fascinating case study in the link between electronic structure, thermodynamic stability and chemical reactivity.
Latest Posts
Latest Posts
-
A Group Of Cells That Perform Similar Functions
Apr 04, 2025
-
How Is Weathering Different From Erosion
Apr 04, 2025
-
What Is The Least Common Multiple Of 12 And 5
Apr 04, 2025
-
Cross Sectional Area Of Cylinder Formula
Apr 04, 2025
-
4 Main Types Of Financial Markets
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Explain Why The Noble Gases Are Unreactive . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
