Drawing Of Law Of Conservation Of Mass
Juapaving
Apr 03, 2025 · 7 min read
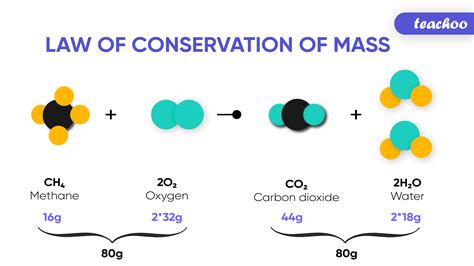
Table of Contents
Drawing the Law of Conservation of Mass: A Visual Guide to Chemical Reactions
The Law of Conservation of Mass, a cornerstone of chemistry, states that matter cannot be created or destroyed in a chemical reaction. This means that the total mass of the reactants (the starting materials) must always equal the total mass of the products (the substances formed). While seemingly simple, understanding and visualizing this law is crucial for mastering stoichiometry and predicting the outcomes of chemical processes. This article will explore the law of conservation of mass through various visual representations, helping you grasp its significance and applications.
Visualizing the Law: From Simple Diagrams to Complex Equations
The best way to understand the law of conservation of mass is through visual aids. These illustrations can range from simple diagrams to complex chemical equations, each offering a unique perspective on the concept.
1. Simple Pictorial Representations
For beginners, simple pictorial representations are highly effective. Imagine a closed system, like a sealed container, containing reactants. These reactants are depicted as distinct shapes or colors, clearly representing different elements or compounds. As the reaction proceeds, the shapes rearrange themselves to form new products, but the total number of shapes remains the same. This visually demonstrates that matter is neither gained nor lost.
Example: Consider a reaction between two elements, A and B, forming a compound AB.
- Reactants: Three A atoms (represented by three red circles) and two B atoms (represented by two blue squares) are present in the container.
- Reaction: The red and blue shapes combine to form AB molecules (perhaps represented by red-blue linked pairs).
- Products: The total number of atoms remains the same – three red and two blue. They have simply rearranged themselves. No atoms have disappeared or appeared magically.
This simple representation clearly illustrates the fundamental principle: the total number of atoms (and therefore mass) remains constant throughout the chemical reaction.
2. Using Ball-and-Stick Models
Ball-and-stick models offer a more sophisticated visual approach, particularly useful for representing molecular structures. Each atom is represented by a colored sphere (ball), and the bonds between atoms are represented by sticks. This model allows for a more detailed visualization of the reaction process, showing how atoms rearrange themselves to form new molecules.
Example: The combustion of methane (CH₄) can be visualized using ball-and-stick models.
- Reactants: Methane (CH₄), represented by a carbon atom (black ball) bonded to four hydrogen atoms (white balls), and oxygen (O₂), represented by two oxygen atoms (red balls) double-bonded together.
- Reaction: During combustion, the bonds in methane and oxygen break, and new bonds form to create carbon dioxide (CO₂) and water (H₂O).
- Products: Carbon dioxide (CO₂), represented by a carbon atom double-bonded to two oxygen atoms, and water (H₂O), represented by two hydrogen atoms bonded to one oxygen atom. The total number of atoms of each element remains unchanged – one carbon, four hydrogens, and two oxygens.
This visualization is far more informative than simple circles, helping to understand the structural changes involved in the chemical reaction.
3. Chemical Equations: The Symbolic Representation
Chemical equations provide a symbolic representation of chemical reactions. They use chemical formulas to represent the reactants and products, along with coefficients to indicate the relative amounts of each substance. A balanced chemical equation is crucial in illustrating the law of conservation of mass.
Example: The reaction of hydrogen gas (H₂) with oxygen gas (O₂) to produce water (H₂O) is represented by the following balanced chemical equation:
2H₂ + O₂ → 2H₂O
This equation demonstrates conservation of mass:
- Reactants: Two molecules of hydrogen (4 hydrogen atoms) and one molecule of oxygen (2 oxygen atoms).
- Products: Two molecules of water (4 hydrogen atoms and 2 oxygen atoms).
The number of each type of atom remains the same on both sides of the equation, ensuring that the total mass is conserved. An unbalanced equation would imply the creation or destruction of matter, violating the law of conservation of mass.
4. Advanced Visualizations: 3D Molecular Modeling Software
For more complex reactions involving large molecules and intricate structural changes, 3D molecular modeling software can provide powerful visualizations. These programs allow users to build molecules, rotate them in three dimensions, and simulate reaction processes, offering an unparalleled level of detail and understanding. These sophisticated tools allow for interactive exploration of chemical reactions, further cementing the understanding of the law of conservation of mass.
Beyond Simple Reactions: Considering Phase Changes and Open Systems
The law of conservation of mass applies to more than just simple chemical reactions. However, its application can be more nuanced when considering phase changes and open systems.
Phase Changes: A Matter of Arrangement, Not Mass
Phase changes, such as melting, boiling, freezing, and condensation, involve changes in the physical state of a substance but not changes in its mass. Ice melting into water, or water evaporating into steam, demonstrates this principle perfectly. While the arrangement of water molecules changes, the total number of water molecules (and thus the total mass) remains constant.
Open Systems: The Challenge of Mass Exchange
Open systems, unlike the sealed container examples we've discussed, allow for the exchange of matter with the surroundings. In an open system, the law of conservation of mass only applies if we carefully account for all mass entering and leaving the system. For instance, if a reaction takes place in an open container and some gas escapes, the mass of the remaining substances will be less than the initial mass of reactants. This doesn't invalidate the law, but rather highlights the importance of considering the entire system, including the mass that has escaped.
Applications and Importance of the Law of Conservation of Mass
The law of conservation of mass is not just a theoretical concept; it has profound practical applications across many scientific fields.
Stoichiometry: Quantitative Calculations in Chemistry
Stoichiometry relies heavily on the law of conservation of mass. It allows us to quantitatively predict the amounts of reactants needed and products formed in a chemical reaction. By balancing chemical equations and using the molar masses of the substances, we can perform calculations related to reactant ratios, yields, and limiting reagents. Accurate stoichiometric calculations are fundamental in various applications, including industrial chemical processes, pharmaceutical manufacturing, and environmental monitoring.
Environmental Science: Tracking Pollutant Movement
The law of conservation of mass is also crucial in environmental science. Tracking pollutants, such as heavy metals or pesticides, in the environment necessitates understanding how their mass changes as they undergo transformations or move through different environmental compartments (soil, water, air). Accurate mass balances are vital for assessing environmental impact and developing effective remediation strategies.
Forensic Science: Investigating Crimes
Forensic science utilizes the principles of conservation of mass in various investigations, particularly in analyzing evidence related to explosions or fires. By carefully assessing the mass of the reactants and products involved in these events, investigators can gain valuable insights into the mechanisms involved and potentially reconstruct the events leading to the incident.
Conclusion: Drawing a Complete Picture of Conservation of Mass
Understanding and visualizing the law of conservation of mass is paramount in comprehending chemical reactions and their implications. From simple pictorial representations to complex 3D modeling, various methods exist to visualize the rearrangement of atoms during chemical changes. It's crucial to remember that although the forms of matter change, the total mass remains constant in closed systems. Mastering the law, particularly through visual aids, is key to success in chemistry and related fields, laying the foundation for more complex concepts and applications. Through various levels of visualization techniques—from basic drawings to sophisticated software simulations—a comprehensive grasp of this fundamental law can be achieved. Further exploration of more complex reactions and open systems is encouraged for a complete understanding of this ubiquitous principle.
Latest Posts
Latest Posts
-
Which Is Worse B Cell Or T Cell Lymphoma
Apr 03, 2025
-
Which Of The Following Is Not Matched Correctly
Apr 03, 2025
-
A Ruptured Follicle Is Transformed Into The
Apr 03, 2025
-
List Some Properties For Ionic Compounds
Apr 03, 2025
-
How Many Degrees In A Parallelogram
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Drawing Of Law Of Conservation Of Mass . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
