Difference Between Glycogen Starch And Cellulose
Juapaving
Apr 05, 2025 · 5 min read
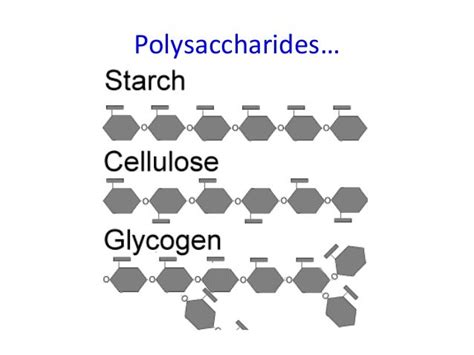
Table of Contents
Decoding the Carbohydrate Trio: Glycogen, Starch, and Cellulose
Carbohydrates are the body's primary source of energy, playing a crucial role in various biological processes. However, not all carbohydrates are created equal. This article delves deep into the fascinating differences between three prominent polysaccharides: glycogen, starch, and cellulose. While all three are composed of glucose units, their structural variations lead to dramatically different properties and functions in living organisms. Understanding these differences is key to grasping their roles in nutrition, biology, and even industrial applications.
What are Polysaccharides?
Before diving into the specifics of glycogen, starch, and cellulose, let's establish a foundational understanding of polysaccharides. Polysaccharides are complex carbohydrates, also known as glycans, consisting of long chains of monosaccharides (simple sugars) linked together by glycosidic bonds. These chains can be linear or branched, resulting in diverse structural and functional characteristics. The most common monosaccharide unit in polysaccharides is glucose, and glycogen, starch, and cellulose are all glucose polymers.
Glycogen: The Animal Energy Store
Glycogen is the primary energy storage polysaccharide in animals and fungi. It's a highly branched polymer of glucose, meaning its glucose units are linked together in a chain with numerous side branches. This branching is crucial to its function.
Key Characteristics of Glycogen:
- Highly branched structure: This extensive branching allows for rapid glucose mobilization. When energy is needed, enzymes can easily access numerous glucose units simultaneously, leading to a quick release of glucose into the bloodstream. This is vital for maintaining blood sugar levels and providing immediate energy for muscle contraction and other cellular processes.
- Storage location: Glycogen is primarily stored in the liver and muscles. The liver glycogen acts as a glucose reservoir for maintaining blood glucose homeostasis, while muscle glycogen provides a readily available energy source for muscle cells themselves.
- Solubility: Glycogen is soluble in water, facilitating its storage and mobilization within cells.
- Digestibility: Enzymes in the digestive system can readily break down glycogen into glucose, making it a readily available energy source for the body.
Starch: The Plant's Energy Reserve
Starch, the major energy storage polysaccharide in plants, serves as their primary source of energy. Unlike glycogen, starch exists in two main forms: amylose and amylopectin.
Amylose vs. Amylopectin:
- Amylose: This is a linear, unbranched chain of glucose units linked by α-1,4-glycosidic bonds. This linear structure makes amylose relatively less soluble in water than amylopectin.
- Amylopectin: This is a highly branched polymer of glucose, similar to glycogen but with fewer branches. The branching occurs through α-1,6-glycosidic bonds, and it's this branching that influences the solubility and digestibility of amylopectin.
Key Characteristics of Starch:
- Mixture of amylose and amylopectin: The proportion of amylose and amylopectin varies depending on the plant source. For example, waxy corn starch is primarily amylopectin, while other starches contain a mixture of both.
- Storage location: Starch granules are stored within plant cells, particularly in seeds, tubers, and fruits. This provides a readily available energy source for germination and growth.
- Solubility: Starch is relatively insoluble in cold water but can form a paste or gel when heated in water, a property exploited in various food applications.
- Digestibility: Like glycogen, starch is easily digested by humans and other animals, providing a significant source of dietary energy.
Cellulose: The Structural Carbohydrate
Cellulose, unlike glycogen and starch, is a structural polysaccharide, forming the primary component of plant cell walls. It's the most abundant organic polymer on Earth. The key difference lies in the type of glycosidic bond linking the glucose units.
Key Characteristics of Cellulose:
- Linear structure: Cellulose consists of long, unbranched chains of glucose units linked by β-1,4-glycosidic bonds. This is a critical distinction from glycogen and starch, which utilize α-1,4-glycosidic bonds. This seemingly minor difference has profound consequences on the properties and digestibility of cellulose.
- Hydrogen bonding: The β-1,4-glycosidic bonds allow for extensive hydrogen bonding between adjacent cellulose chains, creating strong, rigid fibers. This makes cellulose incredibly strong and resistant to degradation.
- Insolubility: Cellulose is highly insoluble in water, contributing to its structural role in plant cell walls.
- Indigestibility by humans: Humans lack the necessary enzyme, cellulase, to break down the β-1,4-glycosidic bonds in cellulose. This makes cellulose indigestible for humans and passes through the digestive system as fiber. However, certain herbivores have symbiotic relationships with microorganisms that produce cellulase, allowing them to digest cellulose effectively.
Comparative Table: Glycogen, Starch, and Cellulose
| Feature | Glycogen | Starch (Amylose & Amylopectin) | Cellulose |
|---|---|---|---|
| Monomer | Glucose | Glucose | Glucose |
| Glycosidic Bond | α-1,4 & α-1,6 | α-1,4 & α-1,6 (Amylopectin) | β-1,4 |
| Structure | Highly branched | Linear (Amylose), Branched (Amylopectin) | Linear, unbranched |
| Function | Energy storage (animals) | Energy storage (plants) | Structural support (plants) |
| Solubility | Soluble | Partially soluble | Insoluble |
| Digestibility (Humans) | Digestible | Digestible | Indigestible |
Applications of Glycogen, Starch, and Cellulose
The distinct properties of these polysaccharides make them valuable in various applications:
Glycogen: While not widely used industrially, glycogen's role in understanding metabolic processes and developing treatments for metabolic disorders is significant. Research focuses on its impact on blood sugar regulation and energy metabolism.
Starch: Starch has numerous industrial applications, including:
- Food industry: Thickening agent, stabilizer, and gelling agent in various food products.
- Textile industry: Sizing agent for yarns.
- Paper industry: Binder and coating agent.
- Bioplastics: Sustainable alternative to petroleum-based plastics.
Cellulose: Cellulose finds widespread applications in:
- Paper industry: Primary raw material for paper production.
- Textile industry: Source of fiber for clothing (cotton, linen).
- Construction industry: Reinforcement material in composites.
- Biofuels: Source of bioethanol.
- Pharmaceuticals: Excipient in various pharmaceutical formulations.
Conclusion
Glycogen, starch, and cellulose, despite all being polymers of glucose, showcase remarkable diversity in their structure and function. These differences are rooted in the subtle variations in glycosidic bond types and branching patterns. Understanding these differences is crucial for comprehending their roles in biological systems and their widespread applications across various industries. Their significance extends far beyond basic biology, impacting areas such as nutrition, medicine, and material science. Further research into the modification and utilization of these polysaccharides continues to open new avenues for innovation and sustainability.
Latest Posts
Latest Posts
-
When A Sperm And Egg Combine It Is Called
Apr 06, 2025
-
How Many Centimeters Is 13 In
Apr 06, 2025
-
How Many Electrons Does An Atom Of Oxygen Have
Apr 06, 2025
-
Are Humans Cold Or Hot Blooded
Apr 06, 2025
-
Words That Begin With A R
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Difference Between Glycogen Starch And Cellulose . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
